For years, zeolite researchers have argued over how surfactants create mesopores in zeolites, key catalysts in oil refining. Now, a new study settles the debate. Raman spectroscopy shows surfactants form micelles inside the zeolite, templating the mesopores from within.
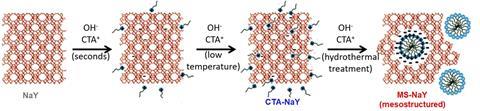
‘Some authors suggested the formation of mesopores involved the dissolution of the zeolite to produce an additional amorphous phase,’ explains Javier García Martínez, from the University of Alicante, Spain, who co-leads the work. However, this proposal didn’t explain some properties of the zeolites, such as their stability and superior catalytic performance. ‘We suggest that individual surfactant molecules penetrate the zeolite crystals and, when the concentration is high enough, micelles form [and] create the mesopores,’ he adds.
However, researchers had only indirect proof of their claims. These included measurements of gas diffusion and porosity, as well as the observation of mesopores using advanced microscopy techniques. ‘None of these experiments [showed] the micelles’ formation, though,’ remarks García Martínez.
To achieve this, they teamed up with Raman expert Maarten Roeffaers at KU Leuven, Belgium, who analysed signature peaks of carbon–hydrogen bonds. These signals correspond to surfactant molecules, revealing their penetration into the zeolite crystals. Moreover, ‘certain signal ratios provide information on polarity’, explains García Martínez. Experiments showcase how polarity decreases steeply – a result of the formation of micelles throughout the zeolite.
‘Following the different bands in Raman enables the creation of 3D images of the process … with unprecedented resolution,’ explains Bert Weckhuysen, an expert in heterogeneous catalysis at Utrecht University, Netherlands. ‘This is a very elegant study,’ he adds. ’It clearly demonstrates that [surfactant] micelles form the mesopores.’
‘This paper finally brings closure,’ says Jeffrey Rimer, from the University of Houston, US, who specialises in porous materials for catalysis. ‘Some people shared scepticism, because the formation of micelles seems counterintuitive,’ he explains. However, the overall process is energetically favourable. ‘It’s surprising, but the stabilisation energy [of micelle formation] compensates for breaking the covalent bonds in the zeolite.’
To carry out their experiments, researchers synthesised their own large zeolite crystals. But they also tested their method on commercial zeolite samples, demonstrating the micelle formation process is universal, explains Weckhuysen. Rimer agrees the importance of surfactant-templating lies in industrial innovation. ‘It was a landmark discovery for zeolites … with a real impact in the field,’ he says, referring to the $80 million (£59 million) start-up founded by García Martínez.
So far, Raman spectroscopy has only provided still images of the zeolite crystal before and after the formation of micelles. ‘Since water is a weak Raman scatterer, potentially you could get the full “live” video,’ says Weckhuysen. Nevertheless, García Martínez considers their latest evidence strong enough. ‘Micelles form mesopores, case closed,’ he says.
References
G Fleury et al, ACS Mater. Lett., 2022, 4, 49 (DOI: 10.1021/acsmaterialslett.1c00514)
















No comments yet