Symmetrical gold and niobium cluster with unusually short Nb–Nb bond
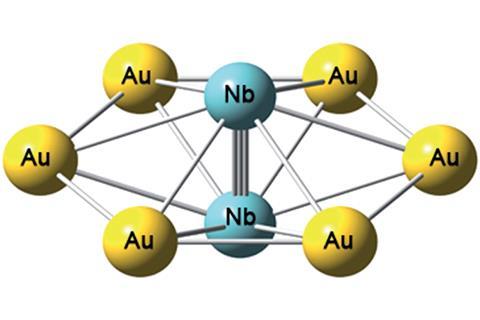
Scientists in the US have made a new molecular wheel. The bimetallic cluster, Nb2Au6-, consists of a Nb≡Nb tripled bonded unit surrounded by a Au6 ring.
Lai-Sheng Wang and his team at Brown University made the cluster by striking a gold and niobium solid target with an intense laser beam. Theoretical calculations show that there are two π bonds and one σ bond in the Nb2 dimer. The cluster also has five totally delocalised σ bonds – scientists have not reported σ aromaticity in a metal–ligand system before.
The antibonding orbitals of the Au6 ring interact with the σ orbitals in Nb2 and reinforce the Nb≡Nb triple bond. So much so that calculations predict the Nb≡Nb triple bond in neutral Nb2Au6 to be 2.076Å, which is shorter than the quintuple bond in gaseous Nb2. Such a short Nb–Nb bond suggests that Au6 rings can serve as robust bridging ligands to enable multiple bonding in transition metal dimers.









No comments yet