An in-situ microscopy technique has allowed chemists to study how gold atoms adsorb on graphene in a range of solvents, helping to increase understanding of solid–liquid interfaces. The researchers hope that this technique will aid the design of better catalysts, fuel cells and batteries.
One of the main ways to visualise individual atoms is by using transmission electron microscopy (TEM), a technique that uses a beam of electrons to image samples. However, this method requires a vacuum, making it challenging to study atoms in solution.

Nanosized cells that trap liquids between two graphene sheets can help get around this issue. Yet, sealing these cells can be difficult, with glue residues often contaminating the interface. Solvents can also evaporate during cell formation, meaning that the concentration of solutions can change, altering the behaviour of atoms at the interface.
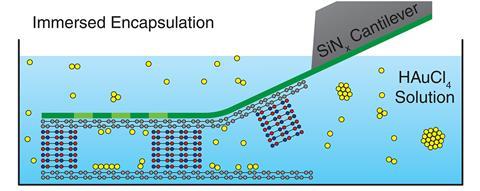
The new method instead submerses cells in solution, where a silicon nitride cantilever then seals the cells with graphene before peeling away ‘like a Post-it note’, explains Sarah Haigh at the University of Manchester, UK, who led the work.
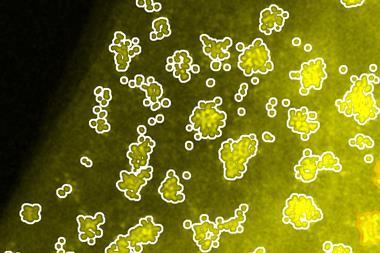
Using this method, the team studied the effect of five solvents – acetone, ethanol, water, butanol and cyclohexanone – on how gold atoms adsorbed onto a graphene surface. An artificial intelligence tool developed by the team tracked and analysed the location of more than 1 million gold atoms in images of the cells.
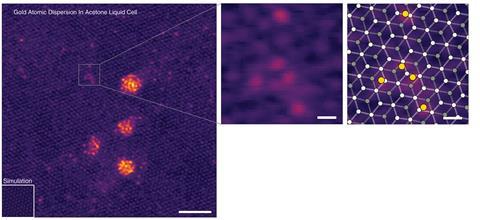
Analysis revealed that in acetone, adsorbed gold atoms were mostly isolated particles. The researchers suggest that this is because acetone’s low polarity leads gold ions to repel each other more strongly in solution. However, in more polar solvents like water and cyclohexanone, gold atoms tended to cluster together in large nanoparticles.
‘Usually you have to infer what’s happening at the solid–liquid interface by looking at the dried [surface],’ explains Haigh. She adds that this technique now allows chemists to directly see what’s happening at the surface.
Alex Roberston, a materials scientist at the University of Warwick who uses TEM, describes the work as a ‘technical tour de force’. ‘[This new] approach gives us understanding as to the nature of atomic interactions that occur with 2D materials immersed in a solution, which are potentially of interest in various catalyst applications.’
In future, Haigh says that ‘it will be super interesting to study a reaction happening at the solid–liquid interface’. She also hopes that this technique will benefit other researchers working on materials with solid–liquid interfaces, such as batteries and fuel cells.
References
S Sullivan-Allsop et al, Science, 2026, DOI: 10.1126/science.adw2469














No comments yet