The first scaffolding approach to making conjugated nanotube architectures has yielded a new family of highly strained carbon-based nanotubes with interesting optoelectronic and electrochemical properties.
Carbon nanotubes come in many shapes and sizes. However, due to the build-up of strain energy involved in bending aromatic systems, building such conjugated and conformationally rigid architectures from the bottom-up has remained a significant challenge to chemists.
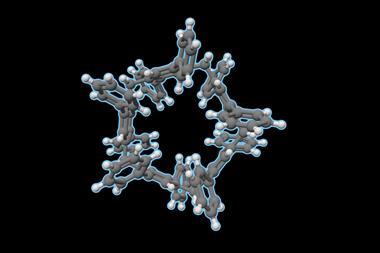
Saber Mirzaei, Edison Castro and Rául Hernández Sánchez from the University of Pittsburgh, US, have now designed a strategy that uses a macrocyclic template to control the direction of the nanotube growth. It starts with a resorcinarene as the base of the container. The process then converts this template into an important octabromo precursor by reacting it with 2,3-dichloro-5,8-dibromoquinoxaline. Due to the intermediate’s preorganised nature, the scientists were then able to form eight carbon–carbon bonds in one step via a Suzuki–Miyaura cross coupling reaction, providing the desired product. Due to the tubular nature of these products, the group named them tubulararenes.
The tubulararenes’ upper rim resembles a cyclo-para-phenylene – the smallest circular segment forming a carbon nanotube. However, as only four of the phenyl rings of this nanoring can rotate in the constrained structure, this ring is even more strained than in free cyclo-para-phenylene.
The researchers established that the tubulararenes are emissive, as expected due to their p-conjugated structure, and they able to accept up to four electrons. Given the large internal cavity (>260A3), host–guest chemistry of these electroactive container molecules is a tantalising prospect for further work.
References
This article is open access
S Mirzaei, E Castro and R Hernández Sánchez, Chem. Sci., 2020, DOI: 10.1039/d0sc03384g

















No comments yet