
A simple hydrogenation reaction can convert challenging-to-recycle rubber wastes into valuable upcycled products. Working independently, two teams from Denmark and the UK exploited the nitrogen functionality in nitrile-butadiene rubber (NBR) to modify the chemical-resistant polymer into an absorbent carbon capture material or stretchy polyol chain. Preliminary studies on real post-consumer plastics – including disposable gloves – gave promising yields of the upcycled polymers and both groups are now looking to scale up their processes.
Nitrile rubber is a staple of any chemistry lab. A copolymer of acrylonitrile and butadiene, it’s flexible, waterproof and most importantly chemically resistant, making it the ideal material for disposable lab gloves – more than 800,000 tonnes are produced annually. However, these very same properties make them almost impossible to recycle. Extensive crosslinking between the polymer chains – known as vulcanisation – prevents mechanical recycling via melting and reshaping. Meanwhile, the inert structure of the polymer chain precludes easy chemical degradation and, as a result, around 99% of nitrile rubber ultimately ends up in landfill.
However, small modifications to the existing polymer structure can transform these properties and two research groups have now developed complementary procedures to valorise this waste plastic.
Activating dormant chemistry
For Troels Skrydstrup and his team at Aarhus University, the end goal was to reengineer this problematic polymer as a carbon capture material.1 Amines are a common motif in solid CO2 sorbents, sequestering the gas via a simple nucleophilic reaction to form a carbonate. ‘Nitrile-butadiene rubber has a nitrogen inside that we would call dormant,’ explains Simon Stampe Kildahl, one of the lead authors on the work. ‘But after a hydrogenation reaction – our activation process – this becomes an active nitrogen, ready to capture the CO2.’
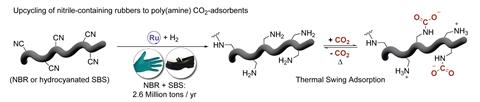
With this transformation from nitrile to amine in mind, the team investigated hydrogenation conditions, heating dissolved stock rubber with a commercial ruthenium catalyst. The optimised reaction converted almost 90% of the acrylonitrile units into the polyamine. But, as carbon capture activity depends on the concentration of nitrogen in the sorbent material, the group wondered whether they could first increase the nitrogen content of their rubber substrates.
Using nickel-catalysed hydrocyanation conditions, they introduced nitrile groups into a proportion of the butadiene units, later repeating the reaction on a second rubber substrate styrene-butadiene-styrene (SBS).
With a sequence established, Skrydstrup’s team next tested the procedure on commercial products, including nitrile rubber gloves and SBS rubber shoe soles. Overall, the reaction tolerated consumer plastics without difficulty. ‘It’s astonishing how well it works considering that it’s a vulcanised material,’ says Clemens Kaussler, fellow lead author.
Polyamine in hand, Skrydstrup’s team turned their attention to the adsorbent properties of the material. The upcycled rubber showed promising affinity for CO2 and the polyamine’s adsorption ability increased with temperature, peaking at 90°C. This contrasts with conventional carbon dioxide sorbents like the metal–organic framework CALF-20, which operates most efficiently at ambient temperatures, and could potentially suit the new material to high-temperature applications like flue gas filtration, says Kaussler.
They proposed this unexpected effect results from the polyamine’s unusual storage mechanism. ‘We can only hypothesise but we have some indications that it is a membrane-like mechanism,’ says Stampe Kildahl. ‘Once you are above a certain temperature threshold, you can achieve both adsorption and also desorption. The CO2 is going like a bucket-brigade from one amine to the other.’
The potential for circularity embedded in this recycling strategy particularly appeals to polymer chemist Marta Ximenis from Polymat in Spain. ‘I think it’s a very elegant approach, showing that with proper catalyst design you can upcycle by modification,’ she says. ‘This is promising for those applications where you really need to valorise or use the CO2, rather than bury it, for example to produce new materials like polycarbonates or even methanol.’
In the short-term, the group are working on developing a scalable catalyst system and improving the reaction’s tolerance towards mixed waste streams but hope the material could ultimately be developed for flue-gas capture applications.
Breaking through the crosslinks
In contrast, for Amit Kumar at the University of St Andrews, the focus was on exploring the potential of nitrile rubber as a feedstock. The group first optimised hydrogenation conditions to reduce nitriles to amines, achieving a quantitative yield with a commercial ruthenium catalyst.2 Incorporating water into the solvent mix and re-optimising the reaction provided a similarly efficient route to the polyol, giving the team two possible products.
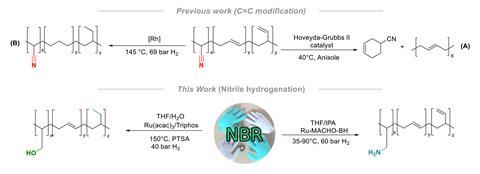
However, translation of these optimised conditions to genuine commercial waste proved slightly more challenging. Vulcanised lab gloves and O-rings were completely insoluble in the reaction solvent. The team therefore employed an aggressive devulcanisation procedure – refluxing in nitrobenzene – which removed the problematic crosslinks and conveniently also completely purified the nitrile polymer. ‘I think for sustainability, this will be our major hurdle,’ says Kumar.
With two upcycled polymers in hand, the group began investigating applications. Preliminary testing showed the polyamine exhibited promising carbon capture activity. Meanwhile, the polyols were unusually stretchable, potentially making them suitable for wound dressing or tissue engineering.
For the immediate future though, Kumar’s focus will be on refining the process to meet the strict demands of industry. ‘These catalyst turnover numbers are still quite low for recycling of plastic waste so this means probably the development of a new catalyst,’ he says.
Despite the scaleability challenges of both methods, these proof-of-concept demonstrations tackle an under-investigated polymer material, says Ximenis. ‘It would be amazing to really bring [these products] back into the supply chain and the next step is exploring how scalable and feasible their processes are to bring them closer to an industrial reality.’
Applicability to real-world systems and processes will be crucial to realise the potential of these approaches says polymer chemist Junpeng Wang from Lehigh University. ‘I think conducting technoeconomic analysis and lifecycle assessment will be the key that can provide feedback on what’s needed to optimise the process to achieve both economic and environmental value.’
References
1 S Stampe Kildahl et al, Chem, 2026, DOI: 10.1016/j.chempr.2025.102918
2 A S L Perez et al, Angew. Chem., 2026, DOI: 10.1002/anie.202525705
















No comments yet