Stack of chiral components creates most enantiopure twisted aromatic ring ever made
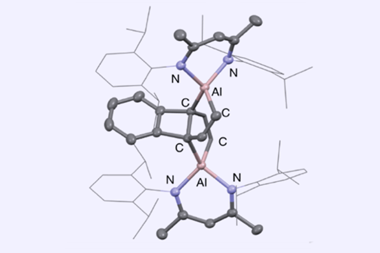
![An image showing (P)-M[28]3·ZnLn](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/480xany/4/7/5/144475_ja-2019-04074d_0005.jpg)
Here is what it takes to make the most enantiopure Möbius aromatic molecule to date: a ring with 30 stereocentres, a set of linkers, zinc ion and its ligand. This chiral amplifier, chemists have discovered, can control a Möbius ring’s chirality, which could be useful for building molecular machines or switchable catalysts.
Möbius aromatics’ Escher-like structure was first predicted in 1964, but it took chemists until 2003 to synthesise the first example. Like antiaromatic molecules, they have 4n π electrons, but the π-orbitals’ half-twist makes them aromatic – and also chiral. But capturing single Möbius enantiomers is difficult since the flexible rings can easily twist into their mirror image form.
In 2017, chemists achieved 76% diastereomeric excess (de) by coordinating a chiral ligand onto the ring to encourage it to twist only one way. Now, scientists in France have upped the stakes, creating a 95% de Möbius aromatic hexaphyrin.
The extended porphyrin is connected to a cyclodextrin with two linkers. Cyclodextrin’s 30 stereocentres are a source of static chirality, which the two linker chains transfer to the Möbius ring by making it planar chiral. But even combined they only create 60% de.
What really amps up the Möbius ring’s asymmetry is a zinc(II) complex. Although both the zinc ion and its ligand are achiral, the way they sit inside the aromatic cavity allows it to only twist one way, producing the highest chiral induction value ever reported for a Möbius ring.
All three parts – the cyclodextrin, the linkers and the zinc complex – can be modified to fine-tune the Möbius ring’s chirality, which the researchers think makes the system promising as switchable asymmetric catalyst or molecular machine.
References
R Benchouaia et al, J. Am. Chem. Soc., 2019, DOI: 10.1021/jacs.9b04074













No comments yet