The study of the life sciences at the subcellular level looks set to be revolutionised thanks to a new microscope that can produce stunning, high-resolution 3D movies of processes ranging from the movement of single molecules within cells to the growth of an embryo. Biologists have described the moving images as ‘truly amazing’.
The new lattice light sheet microscope has been developed by a team led by Eric Betzig, of the Howard Hughes Medical Institute’s Janelia Research Campus in the US. Betzig was a joint winner of this year’s Nobel Prize in Chemistry for his earlier work on super-resolution microscopy.
Light sheet microscopy has been around for several years. It involves illuminating the specimen from the side, sweeping a thin pencil of light, termed a Bessel beam, across the imaging field. The images from that section are recorded, the specimen is moved a tiny fraction and the process repeated. The 2D sections can then be integrated into a 3D image. The process is fast enough to record dynamic events within the sample.
To reduce the time taken to scan a section, Betzig had the idea of dividing the beam into seven parallel parts. As well as obtaining much faster scans, it turned out that the specimens under examination were much healthier: the light energy hitting the specimen was more spread out and ‘gentler’. ‘We were amazed to see a marked reduction in phototoxicity,’ Betzig says.
Lattice of light
In the new work, Betzig and his team have taken things a step further. While the Bessel beams have a highly focused core, they have a less intense region around the periphery of the beam, which creates out-of-focus areas. Betzig pondered the idea of aligning multiple parallel beams: reviving theories he had worked on some ten years earlier he calculated that at a certain, precise spacing of the beams there should be interference that degrades each beam’s blurry edges, and creating a 2D lattice of light.
‘This massively parallel array creates a 2D lattice structure if you have a particular period of symmetry, a magic number,’ says Betzig. ‘But you must hit the magic number with some precision. Even a one percent change will screw up the pattern.’
The team succeeded in putting together a microscope capable of producing the beams that would interfere to create the lattice light sheet. This can scan exceptionally quickly, up to around 1000 planes a second. ‘This is fast enough to cover most of the major things going on in biological systems and also to produce a continuous motion image, rather than a start-stop freeze-frame motion,’ Betzig says.
The images are also much sharper, the light sheet a lot thinner, and the radiation dose bathing the specimen is much softer. ‘Photobleaching is now no longer an issue at all,’ says Betzig.
The team has produced a series of remarkable videos showing, for example, the motion of the microtubule skeleton in a moving slime mould, and the movement of chromosomes and other components in a dividing cell.
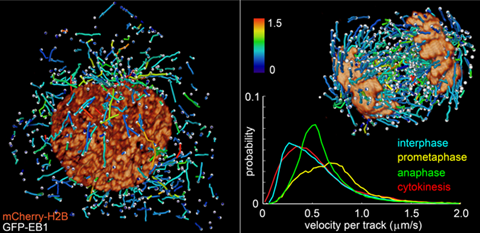
‘One of the most significant aspects of this new system is that it allows us to go from the very small scale, such as single molecule imaging, right up to the development of an embryo,’ explains Betzig. ‘Previously to do super-resolution on single molecules you could only use a very thin specimen. Now we can do this on specimens of arbitrary thickness.’
His co-worker Wesley Legant adds, ‘We know what the microscope can offer in terms of the imaging, but I think there are a lot of applications we haven’t even thought of yet.’
Cells in 3D
Cell biologists are excited by the new microscope. Tomas Kirchausen of Harvard University in the US has, with Betzig’s help, built a ‘clone’ of the microscope in his lab and has used it to film viruses entering cells, for example. Kirchausen says that he can now track processes in a more complete context, visualising individual molecules in a three dimensional space.
Violaine Sée, co-director of the Centre for Cell Imaging at the University of Liverpool in the UK, says the movies produced by the team are ‘truly amazing’. To be able to see such fine detail in complex systems is, says Sée, ‘Fantastic. We see images in our lab every day, but this really is “Wow!”’
Kishan Dholakia, an optical physicist at the University of St Andrews in the UK, says, ‘I think light sheet imaging is an emergent method which could represent a step change compared with other imaging methods in cell and developmental biology.’
He adds that that the high speed of scanning coupled with the low phototoxicity of light-sheet lattices, makes them ‘very exciting’ for imaging applications. ‘I think we may be moving towards an era where light sheet imaging becomes the accepted technique for many areas of study, and this lattice approach has produced some spectacular results,’ he says.
References
B-C Chen et al, Science, 2014, DOI: 10.1126/science.1257998





















No comments yet