Flow process using a protozoan enzyme permits affordable, large-scale inositol production
Researchers in the UK have devised a flow system that produces expensive inositol compounds for a fraction of their current cost. The stereoselective process harnesses the catalytic clout of an enzyme from Trypanosoma brucei – the protozoan parasite that causes sleeping sickness.
When it comes to finding ingenuity in chemical synthesis, you need not look further than the cutting edge in flow chemistry and biocatalysis. In combination, these processes demand both a firm grasp of reaction kinetics and mastery of complex cellular machinery to perform precise tasks. Traditional batch and enzyme-free methods do competently achieve a whole host of transformations, but often lack the specificity to produce the most complex compounds in worthwhile quantities.
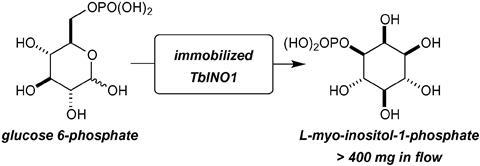
Terry Smith and co-workers at University of St Andrews have employed a biocatalytic in-flow process to produce just such a complex compound – L-myo-inositol 1-phosphate – with unprecedented efficiency. This molecule is the precursor to myo-inositol, a component of various vital biomolecules and the subject of many researchers’ interests. Despite its importance and the low cost of its starting material (D-glucose-6-phosphate) at only €0.03 per milligram, L-myo-inositol 1-phosphate is valued at €196 per milligram. Smith’s group recently observed the extraordinary efficiency of the enzyme L-myo-inositol 1-phosphate synthase in Trypanosoma brucei for producing this molecule and therefore placed it at the core of their synthetic process.
The group first attempted to scale up the reaction under batch conditions, however, Smith explains that they encountered undesirable equilibrium effects when doing so: ‘Upon going above a 10 mg scale in batch form, it soon became apparent that whatever conditions we tried, including recycling the substrate over the immobilised enzyme, we could not get back to 100% conversion.’ Switching to a flow process, by contrast, resulted in near-full conversion at a rate of around 12 milligrams per hour for 36 hours.
‘The researchers demonstrate the potential of a synthase for producing hundreds of milligrams of L-myo-inositol 1-phosphate and demonstrate its utility as a building block for further chemical transformation,’ comments Rob Field, a project leader at the John Innes Centre with an extensive portfolio of work on carbohydrate metabolism. ‘This opens up access to inositol biochemistry in a manner that would be hard to achieve through chemistry alone.’
‘Our method has the potential to greatly simplify access to complex inositol derivatives, and perhaps prompt a new wave of interest in this privileged scaffold,’ says Smith. ‘This includes many useful and important biological molecules that are in high demand to study signalling pathways and numerous biosynthetic routes in a variety of diseases and pathogens.’
References
This paper is free to access
L H Sayer, G J Florence and T K Smith, React. Chem. Eng., 2017, DOI: 10.1039/c6re00175k












No comments yet