Researchers in the US and China have unveiled results in ultracold potassium rubidium dimers, showing for the first time how quantum effects cause deviations from the quantum-statistical model, one of the canonical theories of physical chemistry.
Chemical reactions are fundamentally highly complex quantum transformations of one set of electronic orbitals into another, but the equations required to model reactions on this basis are usually beyond what we can solve. Chemists therefore rely on multiple approximations to determine the path of a reaction.
When atoms or molecules react, the products can be produced in a variety of translational, rotational, vibrational and energetic quantum states. Which state is produced depends on how the energy in the reactants’ states affects the transient intermediate complexes that form for a tiny fraction of a second. These intermediate complexes can have hundreds of different quantum states, many leading to the same product state. A popular assumption in reaction dynamics is that each intermediate quantum state is equally likely. Therefore, the more intermediate quantum states that lead to a specific product state, the more likely that product state becomes.
In their latest study, Kang-Kuen Ni from Harvard University and colleagues cooled potassium–rubidium dimers (KRb) to their quantum ground states – close to absolute zero – in an optical trap. When two KRb dimers collide, they can react to form separate potassium (K2) and rubidium (Rb2) molecules. This reaction is exothermic, allowing it to happen spontaneously even at 500nK. The researchers mapped the statistical distribution of the products’ 57 possible quantum states, correlating products from the same reaction, and looking at where the results deviated from statistical predictions.
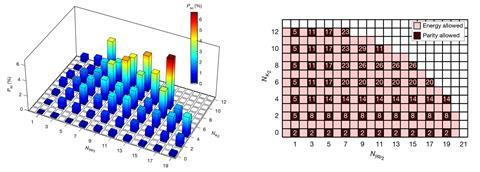
The researchers found that one state in which the products have very high angular momentum was strongly suppressed. They believe this is because, with so much energy going into the products’ rotations, there is insufficient translational kinetic energy remaining for the molecules to escape their potential well and break apart. The reaction, therefore, might never complete. Aside from this extreme example, product states in which the product dimers have high – and very similar – angular momenta were favoured compared with the statistical model. The reasons for this, however, remain uncertain as the team was unable to perform a fully quantum simulation to predict the outcomes.
This is next on their list. The researchers are now planning to study a simpler system in which an atom reacts with a molecule. ‘In that case, exact calculation is more tractable,’ says Ni. ‘If we could achieve the same level of detail in a system in which the theory is tractable, we could see if experiment and theory match.’
Ultracold chemist Matthew Frye of Durham University in the UK believes the work marks a significant step. ‘It’s a wonderful experiment Kang-Kuen has built, and the simultaneous detection of the two product states is quite remarkable,’ he says. ‘It’s allowing us to peel back more onion layers on the chemical reaction, removing layers of averaging: it really is a state-to-state thing now.’
‘Your ordinary lab-based chemist, where most things are in the solid or liquid phase, probably doesn’t encounter it so much,’ says Frye. ‘But in things like atmospheric chemistry and chemistry in space, these theories could be very important.’
References
Y Li et al, Nature, 2021, 593, 379 (DOI: 10.1038/s41586-021-03459-6)











No comments yet