Scientists in Germany have finally resolved the crystal structures of sodium ethoxide along with its corresponding ethanol disolvate.
Sodium ethoxide, C2H5ONa, is a strong base commonly used in organic synthesis, particularly Claisen condensation reactions. Justus von Liebig, widely considered to be the founder of organic chemistry, first reported its synthesis in his manuscript ‘Über die Äthertheorie’ (‘On the theory of ether’) in 1837.
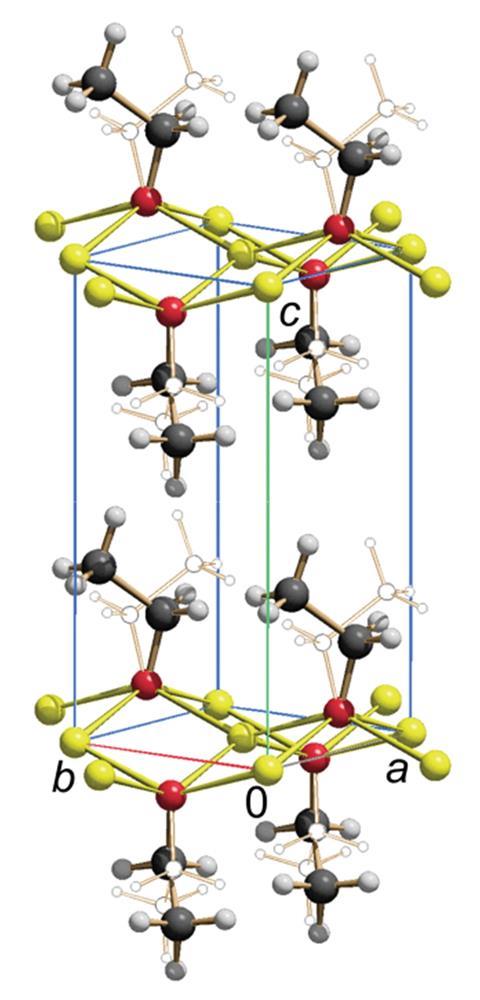
While they were unable obtain single crystals of C2H5ONa, Martin Schmidt and co-workers at Goethe University Frankfurt have now isolated C2H5ONa and analysed its crystal structure using x-ray powder diffraction. They discovered that sodium ethoxide crystallises in the tetragonal space group. It has a layered structure where the sodium ions form a square planar lattice coordinated to four oxygen atoms in a squeezed tetrahedron, with the oxygen atoms situated in the centre of the network, alternatingly shifting above and below each sodium layer. The oxygen atoms are coordinated to five neighbours in a square pyramidal structure, with four sodium atoms in each corner, and an ethyl group at the top of the pyramid.
The C2H5ONa structure is similar to that of lead monoxide. C2H5ONa’s sodium and oxygen occupy the same spaces as oxygen and lead would, in red lead, and its ethyl groups follow the same arrangement as the lead ions’ lone pairs. As a result, the team refers to the structure of sodium ethoxide as ‘anti-PbO type’.

Schmidt and co-workers were able to rationalise the relatively poor solubility of sodium ethoxide in non-polar solvents, such as toluene, because solvent penetrates between and exfoliates the ionic Na–O layers but cannot break them, leaving large layers intact.
References
This article is free to access until 6 April 2020
M Beske, L Tapmeyer and M Schmidt, Chem. Commun., 2020, DOI: 10.1039/c9cc08907a












1 Reader's comment