Chemists have isolated the first homoleptic titanium(0) and zirconium(0) complexes that are seven‑coordinate and stabilised entirely by monodentate gallium ligands. Cooperative gallium–gallium interactions underpin the compounds’ unprecedented stability and may offer a new route to other highly-reduced early transition metal systems.
Early transition metals such as titanium and zirconium are highly electropositive and oxophilic, which means they are most stable in high oxidation states. Attempts to push them into a zero-valent form typically yield fleeting, highly reactive species that require substantial stabilisation.
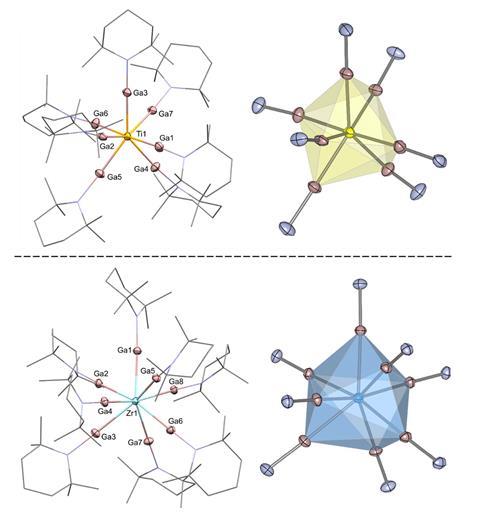
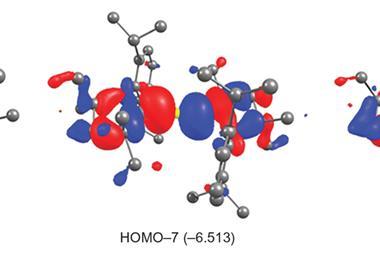
In this new work, that stabilisation is achieved by surrounding the metal centres with seven monovalent GaTMP ligands (TMP = 2,2,6,6-tetramethylpiperidinyl). Each gallium atom, with its diffuse 4p orbitals, accepts strong π-backbonding from the electron-rich metal, redistributing electron density in ways conventional ligands cannot. At the same time, the gallium centres also engage in gallium–gallium covalent interactions, forming a network of tangential, stabilising contacts across a ligand sphere. This cooperative bonding offsets steric repulsion between the bulky ligands, providing the stabilisation required for these unprecedented complexes.
The structures were characterised by single-crystal x-ray diffraction, NMR, IR/Raman, UV–vis and high-resolution mass spectroscopies, while density functional theory calculations revealed the gallium–gallium interactions and electronic features underpinning their stability.
Beyond expanding the chemistry of early transition metals, the work highlights the power of metalloligands as cooperative frameworks. The researchers behind the work propose that the strategy could be extended to capture other elusive low-valent species across the periodic table, including highly reduced f-block complexes.








No comments yet