Millimolar concentrations of water inadvertently absorbed from the air into a solvent were enough to tip the thermodynamic balance between two polymorphs of a supramolecular monolayer, new research shows. By tracing apparently spontaneous structural differences back to a solvent’s history, the work is a reminder that trace impurities can have outsized effects and shows how solvent handling can create reproducibility issues.
Crystal polymorphism is where a compound can crystallise in more than one arrangement. Researchers are keen to deepen their understanding of this structural phenomenon: beyond academic curiosity, it has serious implications for drug safety and efficacy.
The principles governing polymorphism in self-assembled interfacial monolayers are likewise only partially understood. While solvent, solute concentration, temperature and surface are known to influence which monolayer polymorph emerges, researchers occasionally encounter spontaneous polymorphism, in which an unexpected structure appears without any obvious cause. Markus Lackinger at the University of Munich in Germany and his colleagues ran into exactly this problem while studying monolayers of trimesic acid (TMA) on graphite.
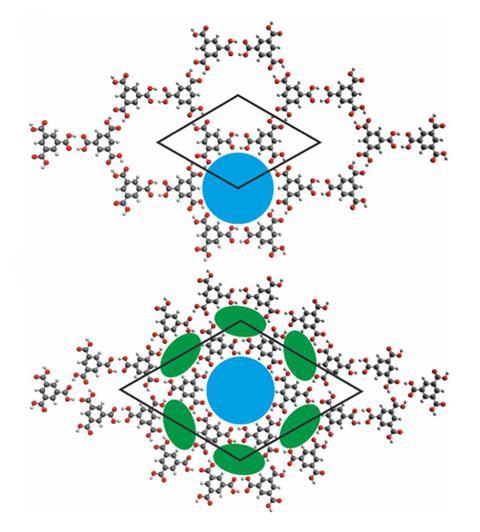
In earlier work, the team had shown that the structure adopted by a single layer of TMA on graphite depends on the solvent used: short‑chain fatty acids favour a dense ‘flower’ pattern, whereas longer‑chain solvents give rise to a more open ‘chickenwire’ network.2 However, when they later attempted to perform similar experiments, the system behaved very differently.
‘We’ve had occasional incidents in which we could never reproduce our own results, [but] these are normally solved by starting from scratch,’ comments Lackinger. ‘We wanted to look into polymorphism on other graphitic surfaces like graphene and intercalated graphite and we bought a new batch of solvent [heptanoic acid], but we only saw the flower structure; this bothered us because we wanted to do experiments with the chickenwire structure. So, we bought another bottle – same thing!’
Seeking an explanation, the team used scanning tunnelling microscopy alongside UV–vis absorption spectroscopy and gas chromatography–mass spectrometry to investigate how the concentration of water in heptanoic acid affected monolayer polymorphism on graphite. They observed that the proportion of the flower polymorph increased with increasing water concentration and, combined with molecular dynamics experiments, concluded that the water impurities disrupt the hydrogen bonds in the trimesic acid monolayers. This disruption was more pronounced for the chickenwire polymorph due to its lower surface packing density and saw the flower polymorph dominate at higher water concentrations.
It turned out that the solvent was unintentionally adsorbing trace amounts of water from the air, meaning that bottles from the same supplier could produce different monolayer structures depending on the batch or how long they had been open.
‘Water as a trace impurity is always on the shortlist of suspects when reproducibility problems arise in chemistry,’ comments nanoscience and surface chemistry researcher Fernando Pablo Cometto from the National University of Córdoba in Argentina. ‘What is genuinely surprising, and impressive, is the degree of sensitivity involved. The fact that concentrations in the range of tens of millimoles per litre – barely a whisper of water in an organic solvent – can completely switch the expressed polymorph is remarkable.’
‘This study is a timely reminder that solvent handling deserves the same rigour we typically apply to our reagents,’ cautions Cometto. ‘Going forward, it makes sense to store solvents more carefully, keep track of how long bottles have been open, and – particularly for concentration-sensitive experiments – move away from the convenience of saturated solutions toward precisely weighed concentrations. These are not dramatic changes, but they are the kind of disciplined habits that make the difference between reproducible science and puzzling irreproducibility.’
Lackinger and co-workers now intend to investigate how water affects the thermodynamic stability of monolayer polymorphs. ‘This is a bit of a special system because the two polymorphs are very close in thermodynamic stability. It would be interesting to see if we can detect the influence of water even if you get the same polymorph,’ concludes Lackinger.
References
1 M Hocke, N Martsinovich and M Lackinger, Nanoscale, 2026, DOI: 10.1039/d6nr00889e
2 O Ochs et al, Chem. Mater., 2020, 32, 5057 (DOI: 10.1021/acs.chemmater.0c00827)














1 Reader's comment