Researchers in Switzerland have found a tripeptide that catalyses carbon–carbon bond formation reactions between aldehydes and nitroolefins with remarkable reactivity and chemoselectivity. The tripeptide retains its catalytic activity in the presence of biomolecules, and in a range of complex mixtures including milk, fruit juice, beer and cell lysates.
Research into peptide catalysts attempts to capture the essential features of enzymatic active sites with just a few amino acid residues. But while enzymes are old hands at catalysing reactions in complex cellular environments, most peptide catalysts have only been tested in pure organic solvents.
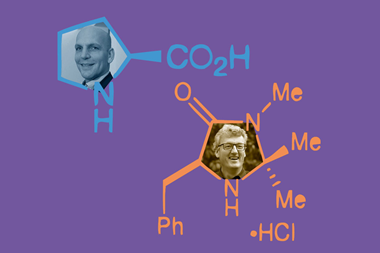
This knowledge gap inspired Helma Wennemers’ team at ETH Zürich to investigate the properties of tripeptide catalysts in complex mixtures. The team started off with an alkylated tripeptide containing two prolines and glutamic acid. They then tweaked the structure, which included adding a methyl group to the central proline residue. The resulting tripeptide displayed remarkably high chemoselectivity that the team attributes to it having a locked trans amide bond.
Wennemers’s team first tested the tripeptide in the presence of biomolecules that could potentially disrupt its catalytic activity including amino acids, nucleic acid derivatives, lipids and vitamins. Next the team set reactions up in a range of solvents that each varied significantly in polarity, pH, viscosity and composition: water from different sources, milk and milk substitutes, fruit and vegetable juices, coffee, tea, honey solution, soft drinks, alcohol, vinegar and olive oil. Despite the challenges, the tripeptide continued to be able to catalyse carbon–carbon bond formation.
‘Our reductionist training often calls for development of catalysts that would work under well-defined conditions, yet in reality, most naturally occurring chemistry happens in extraordinarily complex systems. Thus, catalysts that retain their ability to function in the presence of other molecules are highly prized,’ comments Ivan Korendovych, who researches biocatalysis and chemical biology at Syracuse University in the US. ‘This is a quite exciting validation of the broad applicability of peptide catalysts that will likely foster their further development and, more broadly, will encourage creation for testing of the already available catalysts in complex environments.’
Finally, the team put the tripeptide to the ultimate test: catalysing the reaction in fluid obtained when a cell membrane has been disrupted, which contains intercellular materials such as DNA, RNA, protein and organelles. ‘The findings show the utility of peptide catalysts for organic synthesis and provoke the question of the potential role of small catalytic peptides in the evolution of enzymes,’ explains Wennemers. ‘Furthermore, cells contain a large number of peptides with yet unknown functions. The presented data spark the question of whether some of these peptides catalyse biochemical reactions, orchestrating an underworld of clandestine organocatalysis that yet needs to be discovered?’
Ana Pina, from ITQB NOVA in Portugal, whose research focuses on bioinspired peptide chemistry, says the work provides substantial insights into the impact of the environment of complex mixtures in catalysis. ‘I found this work very exciting because it emphasises the importance of molecular interactions between the microenvironment and peptide catalysts, an unexplored approach that provides valuable directions about evolving and controlling chemical function in stereoselective peptide catalysis.’
References
This article is open access
T Schnitzer, J Rackl and H Wennemers, Chem. Sci., 2022, DOI: 10.1039/d2sc02044k












No comments yet