It mimics phosphate, kills cancer cells in the lab and almost changed how we treat diabetes. So why has a vanadium compound never made it to the clinic?
While most chemists will be familiar with vanadium in the context of metallurgy, catalysis and battery technologies, appreciation of its clinical applications is reserved for a much smaller subset of people.
However, the research is there – and has been for some time. There is a small, but very dedicated, pool of scientists spread across the world who see, and have demonstrated, the promise of vanadium-based compounds for treating cancers and parasitic diseases.
Due to their ability to affect signalling pathways, inhibit protein tyrosine phosphatases, enhance and inhibit the formation of reactive oxygen species, and influence metabolism, interest in the medicinal applications of vanadium coordination complexes and vanadium salts has been there for many years.
Initially much of the focus was on type 2 diabetes. There are a few proposed mechanisms but vanadium compounds have been reported to activate Akt signalling, which is central to glucose and lipid metabolism, and tyrosine phosphorylation by inhibiting protein tyrosine phosphatase 1B, which is involved in the insulin-signalling pathway.
However, the field has faced endless hurdles along the way that have significantly reduced vanadium’s appeal.
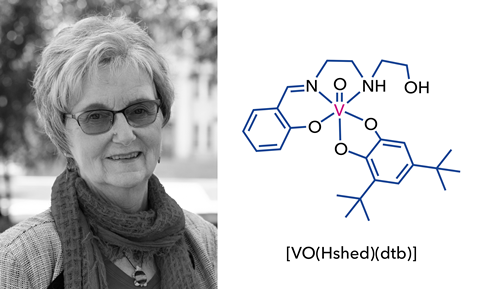
One researcher who has always stood by its side, however, is Debbie Crans. Based at Colorado State University in the US, Crans’ four decades of interest in the fundamental chemistry and biochemistry of vanadium has earned her the unofficial title of the ‘Queen of vanadium’.
She was drawn to the field while working on enzymes that process molecules containing a phosphate and realising the important role that phosphatases plays in biology – ‘it’s the most important way that you can change signalling in a cell; if you change the ratio of phosphatases to kinases, it can influence if you get cancer or not, whether you cure diabetes or not,’ she explains.
It was during this period of fascination with phosphates that she came across two studies , one that showed that you could easily form vanadate esters that mimicked phosphate and the other that showed that vanadate esters were accepted by an enzyme catalysing the oxidation of glucose-6-phosphate. Once vanadium had her attention, she was ‘hooked’.
‘I was interested in the fact that vanadate could mimic phosphate and could replace phosphate… After all, phosphate is essential for life,’ she explains.
‘As a first row early transition metal, vanadium is small and versatile – it can do everything. It doesn’t matter what the enzyme is. It can do redox, it can be a structural analogue of phosphate and of transition states of phosphate ester hydrolysis, it can do all these different things.’
Vanadium and diabetes
It was soon after Crans entered the world of vanadium, in the late 1980s and early 1990s, that Chris Orvig, a medicinal inorganic chemist at the University of British Columbia (UBC) in Canada, and his colleague, John McNeill, a pharmaceutical scientist at the university, were investigating the insulin-enhancing effects of vanadium, sparking huge excitement in the field.
The first report of a vanadium-containing compound – sodium metavanadate (NaVO3) – being assessed clinically for use in the treatment of human diabetic patients was over 100 years ago, in France, in 1899. The researchers first tried the drug on themselves, before testing it on 60 patients (of which three were diabetic) in what they described as a ‘phase 0’ clinical trial. However, interest in vanadium then waned with the discovery, and subsequent adoption, of insulin as the treatment of choice for diabetes in 1922.
Curiosity in vanadium picked up again in the late 1980s and early 1990s. During this time Orvig and McNeill designed and synthesised a vanadium complex of the food additive maltol (BMOV) – and its ethylmaltol analogue (BEOV) – as well as other ligands, and again proposed using vanadium compounds as potential long-term treatments for diabetes.
These compounds showed great promise as insulin-enhancing agents – with phase I and IIa studies returning positive results. However, sadly the economic landscape was against the researchers and, in September 2011 their work halted.
This was a huge blow for Orvig and McNeill, and for vanadium. What had seemed an extremely exciting area of work had been wiped out overnight. It’s now been over 30 years since they published their work on BMOV and BEOV and no vanadium-based compounds have reached the market.
‘Chris Orvig did a great thing for vanadium, because he’s a prominent researcher and amazing communicator,’ says Crans. However, she adds that since their disappointment with BMOV and BEOV, vanadium compounds for diabetes fell and then ‘didn’t move on’.
Funding struggles
One of the key challenges in the field is that, despite there being a lot of potential for vanadium to be used in medicines, funding so far has been extremely limited.
‘[If] you look at the clinical trials of all the metal-based elements – vanadium is on the bottom,’ Crans says. In 2020, Crans published a paper with her then PhD student Kateryna Kostenkova analysing the biological roles of the first-row transition metals.
‘The first-row transition metals are really cool – there are 10 of them and five of them are essential [to human health],’ explains Crans.
‘Then you have three that have biological activity: chromium, that can be very toxic but can also be very beneficial; there is nickel that has a lot of allergenic effects but is an essential element for some bacteria in the biome; and then there’s vanadium that has this amazing impact on signal transduction .’
Researchers have long considered whether vanadium is an essential element for humans. It is certainly essential for some organisms such as ascidians, polychaete worms and Amanita mushrooms. And its ability to interfere with phosphatases, phosphorylases and kinases in humans certainly suggests it has a biological role. However, no deficiency symptoms have been reported, so consensus has yet to be reached.
Crans believes that for many researchers, their caution around vanadium is down to concerns around toxicity. Vanadium pentoxide – which is used as a catalyst – is a toxic pollutant causing multiple respiratory issues and systemic effects.
But when it comes to medicines, Crans points out that there are far more potent metals, such as platinum, palladium and aluminium, that are already used routinely, for example in chemotherapy drugs or in drug formulations.
‘Cisplatin is still used in the clinic – it makes me want to cry; we have vanadium compounds that are 10–100 times more potent and we cannot explore their use for financial reasons. However, as a breast cancer survivor, I know first-hand that drugs like oxaliplatin are still used and save people’s lives.’
The sentiment is echoed by Isabel Correia, an expert in bioinorganic chemistry and coordination chemistry at the University of Lisbon in Portugal.
‘One of the key questions is to convince the community that this toxicity associated with metals makes no sense; toxicity is not inherent, it is a question of concentration for any compound. Metals are traditionally associated with toxicity – [but] this is due to the handling in many sectors, like industry and mining, in which the exposure to metals was a big, big problem.’
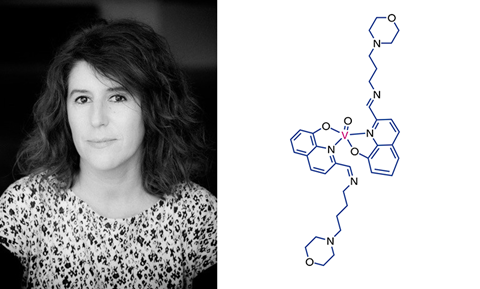
She explains that for many of the heavier metals, the main issue is their tendency to interact with thiolates, such as cysteines, which can induce toxicity, but for vanadium this represents a less substantial problem.
‘In terms of systemic toxicity, vanadium is better than many other heavier elements that might bind to cysteines,’ Correia adds.
Vanadium in medicine
A quick introduction to the chemistry behind vanadium’s clinical potential

Vanadium can assume many oxidation states ranging from -2 to +5 with the main oxidation states being +3, +4 and +5 and is unique among the first row transition metals for its ability to interconvert between multiple oxidations states under physiological conditions where it primarily toggles between V(V) (often in the form of vanadate) and V(IV) (often in the form of vanadyl).

Vanadate, [H2VO4]-

Vanadyl, [VO(H2O)5]2+
Vanadium is not classified as an essential element for humans, despite occasional speculation to the contrary, and deficiency causes no known health issues. A number of teams are studying whether vanadium-based compounds could have useful therapeutic effects.
Current research is examining several ways in which vanadium compounds may interact with biological systems…

Phosphate mimicry: Vanadate is a structural and electronic analogue of phosphate, allowing it to bind in phosphoryl transfer enzyme active sites and inhibit phosphatases and other phosphorylases. This is the basis for its insulin-mimetic effects and its modulation of cancer-relevant signalling.

Redox activity: Depending on oxidation state, ligands and speciation, vanadium can promote or suppress reactive oxygen species, influencing lipid peroxidation and oxidative stress responsible for many antioxidant activities.

Signal transduction: Vanadium compounds can turn certain cell-signalling pathways on or off. Through these changes, vanadium compounds can halt cell proliferation, induce programmed cell death and alter cell movement. These cellular effects underpin their potential as anticancer agents.
Most vanadium complexes hydrolyse or transform in media and in cells. This means the pharmaceutically active species often is not the same as the applied compound – a design and regulatory hurdle that is currently being addressed in multiple ways using novel approaches to drug administration.
The anticancer application
Correia started working with vanadium during her PhD at the end of the 1990s when much of the focus, medicines-wise, was on its role as an insulin-enhancer. However, many of the vanadium compounds under development were also being tested in cell studies as anticancer agents; vanadium salts were first reported to have anticancer properties in 1965.
Since then, the vanadium anticancer complexes that have progressed are structurally diverse and can be used to treat different types of cancer. In recent years, interest in the anticancer applications of oxovanadium(iv/v) and dioxovanadium(iv/v) Schiff base complexes has seen the most growth.
Much of Correia’s work has been with copper, but she says she has always studied the same systems with vanadium.
‘My experience is that, at least in terms of in vitro assays, vanadium complexes are as good or even better than copper ones.’
Vanadium can exist in several different oxidations states , although, physiologically, the two key ones are vanadium(iv) and vanadium(v).
Vanadium(v) species – most commonly in the form of vanadate – are structurally and electronically analogous to phosphate, which allows them to compete for phosphate-binding sites. Importantly, vanadate can mimic the transition state of phosphoryl-transfer reactions, enabling inhibition of phosphate-dependent enzymes. This means it can be used as an inhibitor of protein tyrosine phosphatases to control crucial cell signalling across a variety of different pathways – including MAPK/Erk and PI3K/Akt – disrupting cell metabolism and inducing apoptosis.
‘[Vanadium] can increase reactive oxygen species generation,’ says Correia. ‘[And] it can target the mitochondria, which is really important in terms of anticancer effects.’
As a synthetic chemist, Correia focuses on developing new compounds, particularly by designing and studying ligands. She works a lot with hydroxyquinolines, which have long been recognised to have significant potential in the field of cancer treatment due to their potent chelating properties, stability and broad spectrum of biological effects. When coordinated to a metal centre – such as vanadium – potent therapeutic properties, synergistic or additive effects can be unlocked, including anticancer effects.
‘The strategy is, if the compound reaches the cells in its intact, complexed form, it may exert activity directly; alternatively, it may release the bioactive ligand. In either case, the overall effect is potentially synergistic, with multiple mechanisms contributing to cell death.’
We are not a big community, but we are a very passionate community
The oxidation states of vanadium are very relevant biologically, however, Correia says they also mean that not everyone is comfortable working with vanadium because it requires knowledge of its coordination/aqueous chemistry. Vanadium(iv) is also paramagnetic, which means you cannot study it with NMR, so some researchers prefer working with vanadium(v).
‘People have a tendency to work in fields where they have acquired knowledge. If you take a course on bioinorganic chemistry, you will learn about platinum, you’ll learn about copper, and all the endogenous metals, their processes, [but] vanadium, people don’t learn about it,’ she says.
‘If you then start working with it, you get passionate about it; we are not a big community, but we are a very passionate community.’
Vanadium against parasites
Another clinical area of vanadium research is looking at the role of vanadium in antiparasitic agents.
Dinorah Gambino, a professor of inorganic chemistry at the University of the Republic in Montevideo, Uruguay, and her group are trying to develop metal-based prospective drugs against neglected tropical diseases.
‘We are interested in drugs against diseases produced by protozoan parasites; Trypanosoma cruzi, the causative agent of American trypanosomiasis, or Chagas disease; Trypanosoma brucei, the agent that produces African trypanosomiasis; and finally, Leishmania which causes leishmaniasis.’
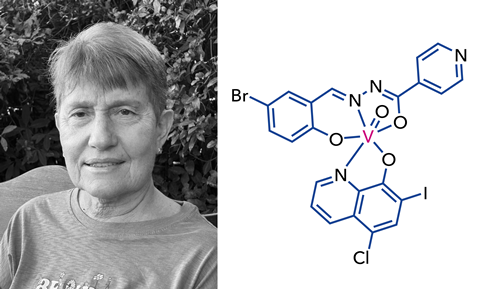
Gambino and her group have performed the first ‘omics’ studies for metal-based compounds in trypanosomiasis to help them unpick the different routes affected in the parasite.
‘Our strategy is a pharmacologically relevant metal, with suitable ligands that coordinate to the metal centre, and we try to get additive or synergistic effects between the ligand and the metal.
‘Vanadium has been suitable for this strategy because of the chemical diversity that vanadium compounds can produce; vanadium has different oxidation states, so it can coordinate to different types of ligands.’
Gambino has faced challenges because of vanadium’s speciation in different biological environments; under physiological conditions vanadium will form different species in response to factors such as pH, ligands and redox environment. ‘In a biological medium, they interchange, and so we have chemical modifications of the compounds when inside a cell or in a culture medium. This makes it more difficult to characterise the active species of our drugs.’
‘Speciation is a difficult topic to study,’ says Gambino. ‘It takes time to study it.’
Her group is working on strategies to make vanadium-containing compounds more suitable for administration – for example using nanocarriers to take the compound to a specific site in the body to help improve overall safety and ensure the drug makes it to its target site without in-body speciation happening.
There is also further work going on elsewhere to investigate advanced delivery systems to unlock the safer clinical use of vanadium, for example incorporating vanadium-compounds into liposomes, which has found that they still retain, or even improve, their biological activity while helping with potential toxicity and biodistribution issues. The use of lipid nanoparticle carriers and intratumoral injections will also be key for anticancer applications.
Fairly recently, researchers have explored using graphene quantum dots as a potential delivery system for vanadium compounds to improve drug solubility in water and membrane transport.
The future of vanadium
For now, the vanadium research community remains small, but those within it are optimistic about its future.
‘I feel like [vanadium] has been around the corner the whole time [I’ve been in academia],’ says Craig McLauchlan, a chemistry professor at Illinois State University, in the US, who has a keen research interest in vanadium coordination chemistry. ‘It just seems that sort of thing that’s percolating in the back.’
McLauchlan says the demise of Orvig’s work had a significant impact on the community: ‘That was so exciting for the whole vanadium community. And then it was just like “nevermind” – [it] really caught us all out.’ But, he believes that in the past five to 10 years conversations around vanadium have been building momentum again.
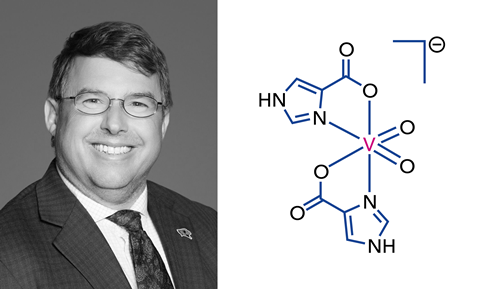
‘It’s been exciting to see vanadium pick up in some of the antiviral and anticancer areas, because now you’re talking about things that are more acute.’
Adnan Zahirović, an inorganic chemist at the University of Sarajevo, in Bosnia and Herzegovina, believes the field is ‘scientifically very fertile’.
‘Many of the people who shaped the field of vanadium in medicine – people like Debbie Crans, Chris Orvig, McNeill, [etc] were my entry point into this area. When you read their work and look at the consistency of the data over decades, it is very hard not to be inspired: vanadium complexes show a remarkable spectrum of biological activities and very robust insulin-mimetic and insulin-enhancing effects in vitro and in vivo.’
He strongly believes that it is important for the next generation of researchers to continue to build on vanadium chemistry in a medical context; his personal goal is to use modern coordination chemistry and bioinorganic tools to redesign and optimise vanadium complexes to address the key limitations that have held them back in the past.
‘I am particularly interested in rational ligand design and in understanding speciation and targeting at a mechanistic level, so that we can deliberately “tune” vanadium complexes towards safer, more selective and clinically realistic candidates,’ he says.
However, he acknowledges that there is a reason why we still do not have a ‘vanadium pill’ on the market. From concerns around long-term accumulation and organ toxicity and the complexity of speciation under physiological conditions, he explains: ‘[it] makes it challenging to define a clean drug entity that regulators and [pharmaceutical] companies are comfortable with’.
As a result, he doesn’t think the most realistic future scenario is a simple, chronic, high-dose oral vanadium drug for type 2 diabetes, but perhaps a role in ‘more refined, second-generation applications’.
‘Carefully designed coordination compounds with improved stability and biodistribution, targeted or nanoformulated systems, or combination approaches where vanadium is used together with antioxidants, flavonoids or even other metals to mitigate toxicity and enhance efficacy.’
For Crans, retirement is not on the cards just yet – she is excited by new reports showing that properly formulated vanadium compounds can be more active and less toxic for treating diabetes. She is also pursuing a new approach to tackling substance abuse, specifically cocaine addiction.
‘It is wonderful to recognise that at least some metal‑based compounds are now being explored as therapeutics,’ concludes Crans. ‘As we continue to develop drugs and diagnostics, it is important to recognise that small molecules have advantages that can be very beneficial – and that we need to take advantage of far more chemical space than has been used in past decades.’
Correction: The formula for vanadyl was corrected on 2 June 2026

Vanadium’s promise in medicine and the researchers who refuse to give up

It mimics phosphate, kills cancer cells in the lab and almost changed how we treat diabetes. So why has a vanadium compound never made it to the clinic?
 Currently
reading
Currently
reading
Vanadium’s promise in medicine and the researchers who refuse to give up
- 2


















No comments yet