Unusual ionic conducting framework retains conductivity at −20°C to open up a promising class of electrolytes
Ionic liquid conductivity decreases at low temperature. To overcome this, a team of researchers from across Japan have combined an ionic liquid with a metal–organic framework (MOF) to produce an unusual material that retains its conductivity below –20°C. This low-temperature conductivity, together with the attractive ionic liquid properties of non-flammability and negligible volatility, could open up the potential for safe battery and capacitor materials for use in extremely cold conditions.
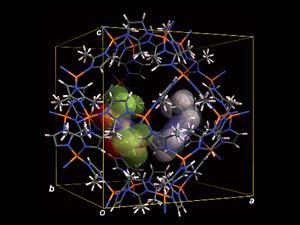
Temperature drops cause ionic liquids to slow down and form an ordered frozen phase. Kazuyuki Fujie, from the Kyocera Corporation R&D centre in Kagoshima, and co-workers demonstrated that a select choice of framework can interfere with ion pair interactions to prevent this phase from forming. ‘It is a challenge to prevent ionic liquids from freezing without chemically modifying the ionic liquid itself,’ says Fujie, but by combining MOF ZIF-8 with the ionic liquid EMI-TFSA (1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)amide) the team have confirmed that the ionic liquid–ZIF structure is more conductive than that of the same bulk ionic liquid below –20°C.
The conductivity of the ionic liquid–ZIF structure is maintained below –20°C due partly to the ionic liquid ion pair arrangement being disrupted by ZIF-8. This phenomenon may be down to the unique structure of ZIF-8, which has cages connected together by two different sizes of windows. ‘The results indicate that conductivity may not be significantly influenced by the interaction of molecules, but by the interaction between the ionic liquid and ZIF framework,’ comments ionic liquid expert Johan Jacquemin, from Queen’s University Belfast in the UK.
Ionic liquid molecules move from the outside cages of ZIF-8, through the bigger of the two window sizes into the inside cages, until equilibrium is reached and the cages host separate pockets of ionic liquid molecules. ‘The window size of the MOF plays a crucial role in the control of the movement from cage to cage,’ explains Jacquemin. Small pockets of ionic liquid molecules across the structure prevent the continuous ordered frozen phase from forming, so the ionic liquid–ZIF structure is disordered, and the ionic liquid in the MOF pores is able to function like a liquid, retaining its conductivity at low temperature. ‘This finding is important for understanding interaction changes and transport properties that occur in a MOF in the absence or presence of ionic liquids. It would be interesting to see how ionic liquid–MOF specific interactions influence ionic liquid properties such as solvation and reactivity,’ says Jacquemin.
As for their potential use in low-temperature power supply, Fujie believes that the ionic liquid–MOF is potentially useful if a better combination of ionic liquid and MOF is found. ‘At this time, an enhancement of the conductivity and reduction in the cost are needed to replace existing technologies.’
References
This paper is open access. Download it here:












No comments yet