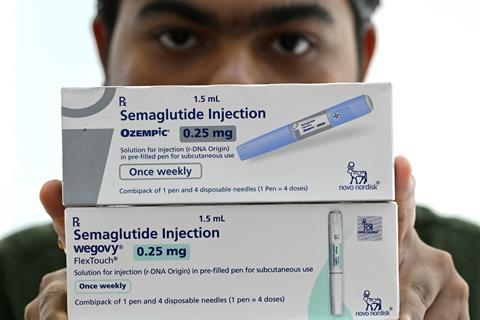
Generic drugmakers in India had 20 March 2026 marked in their calendars for quite a while. That’s the date that Indian patents protecting Novo Nordisk’s semaglutide (Ozempic/Wegovy) expired. Dozens of generic versions of the drugs are now expected to enter the market, at significantly lower prices than Novo’s.
Semaglutide kick-started a transformation in treating diabetes and obesity. Along with Eli Lilly’s related tirzepatide (Mounjaro/Zepbound), these new drugs have stormed towards the top of drug sales lists. They have also stimulated a pipeline of follow-up injectable and oral drugs, as other companies attempt to enter a lucrative market, while Novo and Lilly seek to reinforce their positions as market leaders.
Three factors have combined in India’s rapid introduction of generic semaglutide. First, India’s patent laws make it difficult to extend patent protection on medicines by tweaking processes or formulations. That means Novo’s patents are expiring earlier in India than in many other countries. That said, owing to a remarkable oversight, Novo’s Canadian patent lapsed after the company failed to pay its maintenance fees. But while generic versions could have been introduced in Canada earlier this year, so far, they haven’t appeared.
Secondly, India is home to many of the world’s most successful generic drug manufacturers. Their combined infrastructure and expertise in developing cost-effective manufacturing processes is enormous. Finally, demand for these medications in India is increasing rapidly, owing to growing numbers of people in India with diabetes and obesity – many of whom cannot afford Novo’s prices. That makes for an enormous opportunity for generics.
The bonanza of generic semaglutide medicines in India will likely be accompanied by intensive marketing efforts as the various manufacturers compete for their share. But amidst that clamour there is a significant risk of some producers taking advantage of the proliferation to sell unauthorised versions of the drugs, or using improper marketing practices to gain an unfair – and potentially unsafe for patients – advantage.
Anticipating such potential problems, India’s drug regulator has increased its regulatory surveillance and enforcement around the supply chain for GLP-1 weight loss drugs. In the weeks leading up to the release of authorised generics, the regulator increased inspections at online pharmacy warehouses, drug wholesalers, retailers, and wellness and slimming clinics.
The rest of the world will be watching closely as the landscape unfolds in India, both in terms of price competition and influence on prescriptions of semaglutide medicines compared to Lilly’s tirzepatide. In many countries tirzepatide has been overtaking semaglutide on the back of trial results suggesting marginally greater effectiveness. The most successful Indian manufacturers will also have an eye on rapid expansion into worldwide markets as patents expire in China, Brazil, Turkey and South Africa later this year. Novo’s EU and UK patent protection will last into 2031, and in the US until at least 2032.















No comments yet