Ben Valsler
I like to spend my free time looking through cupboards trying to find products that consist of very few compounds. It’s actually pretty tough, as almost everything consists of a complex mixture. And it seems, unusual as it may be, it’s a hobby I share with Brian Clegg…
Brian Clegg
Should bored chemists decide to spend a wet weekend looking through the contents of their store cupboards at home, they would find few products that depend almost entirely on a single compound. In the food cupboard there’d be the inevitable sodium chloride, but more subtly, amongst the cleaning products, the chances are high that there would be a solution of sodium hypochlorite – because it is better known as bleach.
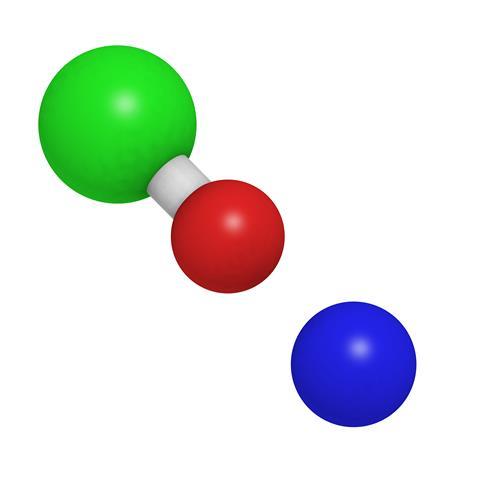
This simple inorganic compound is just NaClO – the only distinction from that common salt is that instead of a chloride ion it has hypochlorite with an added oxygen atom, but the transformation in the substance is striking. Outside of the lab we don’t usually come across anything other than a solution of sodium hypochlorite, but it does form a white powder, which can decompose dramatically when heated, or a yellowish hydrated solid that melts at room temperature.
The common name also gives away the other main practical application of the compound
The primary use of bleach around the house tends to be a disinfectant for toilets and drains, or diluted to make it suitable for sanitising surfaces, though it tends to be used less now as safer alternatives are now available as antibiotic cleaning sprays. The disinfectant effect is both the result of the hypochlorite releasing chlorine, and of forming hypochlorous acid with water – each acting as a very strong oxidising agent, ripping apart proteins and DNA in bacteria and destroying their cell membranes.
The common name also gives away the other main practical application of the compound. The word ‘bleach’ shares its roots with some languages’ words for white (think ‘blanc’ in French), and bleaching is generally about removing colour and stains. Here the hypochlorite either destroys the molecule causing the colour, or changes double bonds into single bonds, which reduces the absorption of visible light, meaning that white light is re-emitted, rather than light of a particular colour. Although sodium hypochlorite is the definitive ‘bleach’ the term is sometimes used for other products: bleached blonde hair, for instance, is traditionally produced by hydrogen peroxide.
However, some of the earliest practical uses of sodium hypochlorite were rather less domestic – specifically in the messy, smelly business of getting from cat and other animal guts to musical instrument strings. In the early part of the nineteenth century, a prize was offered in France for finding a way to separate the appropriate part of the guts without producing a putrefying mess. Chemist Antoine Germain Labarraque picked up on sodium hypochlorite, which had been produced a couple of decades earlier by Claude Louis Berthollet and was already produced by the Javel company as ‘liqueur de Javel’ for bleaching cotton and other materials. Labarraque found that the bleach solution successfully detached the gut membrane and seemed to reduce the odorous decay.

Soon after, on the death of the French king, Louis XVIII, Lebarraque was able to use sodium hypochlorite to reduce the offensive stench from the gangrenous body, which led him to extensively explore the use of such compounds in disinfection and the removal of the odour of decay, establishing the sanitising capability of hypochlorite alongside its established bleaching role.
The compound was originally produced by bubbling chlorine gas through sodium carbonate, but since the twentieth century the only significant production approach, known as the Hooker method, has involved passing chlorine into dilute sodium hydroxide and electrolysing the mix.
There are several thousand domestic accidents with bleach in the UK alone
There is no doubt that sodium hypochlorite has done plenty of good in reducing infection by killing bacteria, from the sink to the swimming pool, where it is often used in pool chlorinators. It turns up everywhere from paper mills to water purification plants both as a bleach and a mechanism to control bacteria, while its oxidising abilities also find it turning up in petroleum refining and as a neutraliser for nerve agents. But it is also one of the most common hazardous materials we experience, both because of the destructive nature of the hypochlorite and the small amount of sodium hydroxide that tends to be used in the solution to help prevent the hypochlorite from breaking down.

There are several thousand domestic accidents with bleach in the UK alone, often caused by children accidentally drinking the water-like solution. What’s more, if bleach meets an acid it will give off chlorine gas, while it reacts with alkaline household cleaners to produce toxic chlorinated volatile organic compounds like carbon tetrachloride and chloroform. But as long as it is treated with respect, sodium hypochlorite will continue to be a compound that brightens up our lives.
Ben Valsler
Science writer Brian Clegg, on perhaps the most common chemical hazard we face – domestic bleach. Next week, a compound beloved of biologists that needs the right atmosphere to really show its stuff…
Kat Arney
In the cold light of day, fluorescein doesn’t look like much – just a dull orangey-red powder. But like a party animal whose true colours only show after a few drinks and a spin on a nightclub dancefloor, dissolve it in alcohol or water and put it under an ultraviolet light and fluorescein comes alive, glowing with an eerie green hue.
Ben Valsler
Find out more with Kat Arney in next week’s Chemistry in its Element podcast. Until then, feel free to get in touch with any questions or comments – email chemistryworld@rsc.org, tweet @chemistryworld or look us up on Facebook. I’m Ben Valsler, thanks for joining me.













No comments yet