Ben Valsler
This week, Katrina Krämer looks for the truth about reactive species known as radicals.
Katrina Krämer

When you start typing ‘are free radicals…’ into a Google search, you get a number of suggestions, including: are free radicals good? Are free radicals bad? And even: are free radicals real? While the latter question is easy to answer – yes, they are definitely real – compounds with unpaired electrons seem to combine good and evil like few other classes of molecules.
There’s a theory that free radical damage to living cells is the sole cause of aging. Although this theory is highly controversial, certain nutrition websites love this idea and have promptly declared radicals ‘our aging enemy’. The antioxidant-rich diets these sites promote supposedly fight radical damage and keep you looking 20 until well into your 80s.
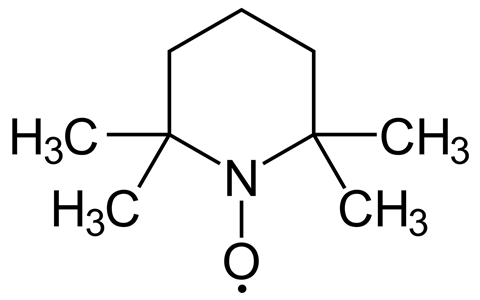
On the other hand, there is evidence that radicals are involved in tissue healing processes. Scientists have also discovered that radicals can be used in medical treatments to target and destroy cancer cells. And then there’s (2,2,6,6-tetramethylpiperidin-1-yl)oxyl – colloquially shortened to TEMPO. One of the most loved radicals, TEMPO has many talents that serve organic chemists, polymer scientists and molecular biologists alike.
Despite its unwieldy moniker, the bright orange solid that is TEMPO is a well behaved lab mate. As a persistent radical, it can be stored for years – unlike some of its more reactive cousins. The hydroxyl radical, for example, has a half-life of just a billionth of a second. The reasons for TEMPO’s longevity lie in its structural and electronic design, which simply don’t allow it to be more reactive – but we’ll get to that later.
Today, TEMPO is a staple reagent. But just over 100 years ago, scientists didn’t even know long-lived radicals existed. Here’s Daniel Allwood, organic chemist at Sheffield Hallam University, UK, recounting the beginnings of radical chemistry.
Daniel Allwood
The discovery of stable radicals coincided with our discovery of radicals in general, because these reactive intermediates have been taking part in reactions all the way throughout prehistory. The first person who discovered a stable radical was Moses Gomberg, who was a professor at the University of Michigan. In 1900 he was trying to prepare hexaphenylethane using a Wurtz coupling from triphenylmethyl chloride. What he actually found was that he had prepared the dimeric peroxide from triphenylmethyl chloride, and he deduced from that that he had prepared a trivalent carbon species which had then reacted with oxygen in the air to give him this peroxide.
Katrina Krämer
The trivalent carbon species Gomberg had made turned out to be the first persistent radical. He reported his finding that same year, concluding the paper by writing ‘this work will be continued and I wish to reserve the field for myself.’
He did indeed become the authority in radical chemistry, but maybe not – at least initially – for the reasons he had imagined. Most of Gomberg’s colleagues thought his work to be of little use. After all, radicals were little more than a molecular oddity. Others simply didn’t believe his findings at all. A radical containing a trivalent carbon atom went against everything chemists knew about carbon at the time, which they thought must always have four bonds.
Only 30 years later, radicals started to make waves. The rise of synthetic polymers fuelled chemists’ interest. These odd molecules and their unpaired electrons allowed them to do a new type of reaction: radical polymerisation.
Scientists were soon looking for a way to bottle up and store radicals’ reactive power. In 1960, TEMPO was discovered by two Russian chemists, Lebedev and Kazarnovskii . They found that not only was it easy to make but also remarkably stable.
Daniel Allwood
Principally, the fact that your radical is centred on an oxygen which is directly connected to a nitrogen means that you’ve got a non-bonding pair of electrons on the nitrogen in the form of a lone pair, and these can resonance stabilise the radical on the oxygen. So by donating the electrons from the nitrogen through to the radical on the oxygen, you form a three-centre two-electron bond, and that resonance stabilisation is what makes TEMPO electronically more stable than a normal radical.
Katrina Krämer
Moreover, the four bulky methyl groups flanking the nitrogen make it almost impossible for TEMPO to dimerise – two TEMPO molecules just can’t physically get close enough to form a bond. The methyl groups also allow the radical to hyperconjugate, sharing the burden of an unpaired electron with empty orbitals.
Though it is difficult to summarise TEMPO’s many talents, chemists might have encountered it as a radical detective.
Daniel Allwood
In our lab, we use TEMPO as a simple radical trap. A lot of the reactions that we’re developing, a lot of the reactions that we’re discovering, we either strongly suspect or we kind of have an idea that they involve radical intermediates, or we’re trying to actively generate radical intermediates. And usually these are transient radicals, they’re reactive radicals because we want them to participate in things like cross coupling reactions.
The issue then is how do you characterise your reactive intermediates, because they exist for such a short amount of time that you would never observe them by any spectroscopic method. So what we do is – if we have a reaction where we think there’s a radical intermediate taking part, we run the reaction again in the presence of an excess of TEMPO, and because TEMPO is a radical and it’s unlikely to react with itself, it reacts with the transient radical, you get combination of the unpaired electrons, and you form an adduct which is diamagnetic – you can isolate it and you can then run standard NMR spectroscopy characterisation techniques.
Katrina Krämer
TEMPO is also a radical mediator that can control its wilder cousins. Added to a polymerisation, TEMPO combines with the active radical at the end of a growing polymer chain and stops it from reacting further. But this new bond is quite weak. A little heat is enough to release TEMPO and restart the polymerisation – a neat trick that allows chemists to precisely control their polymers’ chain lengths.
And there’s more: biochemists and molecular biologists are making good use of TEMPO’s unpaired electron as a molecular flare.
Daniel Allwood

Things like, not so much TEMPO but other nitroxyl radicals, can be used as what we call spin labels. Because TEMPO and other radicals have an unpaired electron, they’re paramagnetic, which means that they can be detected by electron paramagnetic resonance (EPR) spectroscopy. So if you incorporate a spin label into, for example, a biological molecule such as a protein or an enzyme, you can detect where that spin label has been incorporated using EPR spectroscopy. And that allows you to probe the structure and function of certain proteins and enzymes.
Katrina Krämer
It sounds like scientists are making a strong case for TEMPO. But what’s the final verdict on other free radicals: do they hold the key to eternal youth?
Daniel Allwood
Radicals are reactive species, there’s no denying that. And our cells contain a lot of very sensitive cellular machinery, and if you put a reactive species in the presence of cellular machinery it will damage it. But our bodies are very good at dealing with damage. Every cell in our body, every day, undergoes ten thousand instances of oxidative DNA damage. And the vast majority of those are repaired or dealt with in some way.
Radical species probably do contribute to aging and there’s a lot of research which suggests that that is the case. But there are a lot of other factors which are involved in aging. A lot of the animal and clinical models that we’ve run on the process of aging give conflicting results, so in some species we get the opposite results to other species.
So the short, and probably quite disappointing answer is that we’re not quite sure yet, but it’s probably not as simple as just saying it’s exclusively down to free radicals.
Katrina Krämer
So figuring out what role radicals play in the complicated chemical machinery that is our body is a tough task, but, by helping us study biological processes, maybe one that TEMPO will help us to understand.
Ben Valsler
That was Katrina Krämer speaking to Daniel Allwood from Sheffield Hallam University. Next week, Mike Freemantle on the preserving power of fructose.
Michael Freemantle
Only then did I look at the label on the bottle. The ’best before’ date was the end of November 2003. The half full bottle had therefore been in our drinks cabinet untouched for at least 14 years.
Ben Valsler
Join us next time to find out how fructose kept Mike’s drink sweet for well over a decade. Until then, drop us a line with any questions or comments – email chemistryworld@rsc.org or tweet @chemistryworld. I’m Ben Valsler, thanks for joining me.













No comments yet