Rachel Brazil finds out how chemists can restore art to its former glory

The history of art conservation is almost as long as the history of art itself. Michelangelo’s Sistine Chapel frescoes were first restored in the mid-16th century, only decades after being painted. Water damage had caused a white potassium nitrate scale, which was treated with linseed and walnut oil. But such cosmetic solutions were sticking plasters and lacked the scientific understanding that has now revolutionised the field. Today, conservation scientists are finding innovative ways to clean and protect our cultural heritage.
Joyce Townsend, a conservation scientist at the Tate Gallery in London, UK, explains that all conservation projects start with a detailed assessment of the work. ‘There is always a debate between conservators, curators and scientists thinking what is absolutely necessary to be done. Is there an option right now to do nothing at all, or is that unsafe because the object is deteriorating?’ As well as providing for an artwork’s survival, Townsend describes the basic ethical principles of any conservation process as ‘ensuring that you do no harm to the work, and that wherever possible anything you do could be reversed by a later generation’.
Cleaning aims to restore artworks to how the artist intended them to look, but this is rarely simple and sometimes controversial. For example, the most recent Sistine Chapel restoration in 1994 had a small number of critics expressing horror and labelling the now bright frescoes ‘Disney-esque’. Conservation scientist Bronwyn Ormsby, also at the Tate Gallery, believes a more nuanced view is required. ‘The concept of taking a work back to its original appearance is essentially flawed, as materials change with time regardless of the level of protection.’

How an artwork is cleaned will depend on the nature of the material to be removed. With paintings, a variety of organic solvents are used, but the most common solvent is water, often with chelating agents, surfactants or salts to control pH. Applying solutions through tissues, gels and sponges is becoming the norm, due to the level of control offered by holding the cleaning system at the upper surface of the art. Such gels, introduced in the late 1980s, are usually water-based emulsions thickened with cellulose or synthetic polymers. By slowly releasing the solvent, they prevent some of the swelling damage that free solvents cause to paint layers.
Cleaning up past mistakes
The problems that conservators face are sometimes caused by past conservation treatments. Historical conservation techniques seem bizarre to us today – for example, the Sistine Chapel was restored in the seventeenth and early eighteenth centuries using, among other things, wine and sponges made of bread. The common practice of ‘lining’ deteriorating canvases is a good example of a process that now causes problems. ‘In the nineteenth century, they did this by hand ironing on the surface and actually pressing the painting. That has led to a lot of changes,’ Townsend explains. ‘At the same time, if they hadn’t done it, these paintings might not be with us today.’
Conservation does not always take place in a lab
The problem is not confined to the distant past: during the 1960s, for example, it became popular to use synthetic polymers to consolidate and stabilise frescoes – plaster-based wall paintings. They seemed like the perfect replacement for the wax coatings previously used, but over time it became clear that this was not the case. Their presence drastically changed the paintings’ surface properties, causing mechanical stresses and crystallisation of salts beneath the painting leading to accelerated disintegration. In addition, the polymers themselves became discoloured and brittle, particularly in hot countries such as Mexico, where they were extensively used on murals at pre-Colombian archaeological sites.
These problems have driven innovations, but so has the need to create treatments that do not expose conservators to harmful solvents and chemicals. Conservation does not always take place in a lab – a conservator might be treating a fresco in a small, badly ventilated space. Health and safety concerns have motivated new methods, as well as a desire to use green processes that minimize environmental harm.
Destroying dirt with lasers

One method that ticks these boxes is laser ablation. Surface deposits can be removed from artworks with short bursts from a laser beam. The technique was first used in the 1970s for cleaning ‘black crust’ from marble buildings and sculptures. This is a deposit often found in polluted environments and can be difficult to clean. It is largely gypsum (calcium sulfate dehydrate) mixed with airborne particulates. The method was slow to catch on as lasers were very expensive, and there was also scepticism about the cleaning control it offered.
By the mid-1990s, laser cleaning was established for stone and started to be used for other materials such as gilded bronzes and frescoes. A major breakthrough came when an Italian physicist at the National Research Council Institute of Applied Physics in Florence, Salvatore Siano, developed a method that used even shorter pulses, of only micro to nanosecond duration. The neodymium-doped yttrium aluminium garnet (Nd:YAG) lasers commonly used in conservation emit light of 1064nm wavelength, in the near infrared. They are hand-held, portable devices and in most cases treatments will be carried out on a wetted surface to prevent over-heating. Siano’s work investigated a number of pulsing methods, including Q-switching which uses an optical switch to create a maximum energy pulse as short as 6ns.
The laser cleans by heating and expanding the surface, causing pressure waves that act like a rapid vacuum cleaner burst and detach surface material. If water is present at the interface between layers, its vaporisation also produces fractures. Shorter high-intensity laser pulses favour pressure effects while thermal effects dominate with longer pulses. By carefully selecting the pulse energy criteria, the laser will remove encrustations and dirt while leaving the material underneath unharmed.
Siano and his team first used laser treatments on metals in 2001 with a major restoration of the Porta del Paradiso (Gates of Paradise), the gilded-bronze doors of Florence’s Baptistery of San Giovanni, designed in 1401 by early Renaissance artist Lorenzo Ghiberti. The masterpiece, often claimed as the starting point of the Renaissance itself, contains 40 gilded reliefs of Old Testament scenes, which were heavily encrusted. ‘The use of an infrared wavelength, which is highly reflected by gold, makes the laser removal very safe without any risk of damaging the gold film,’ explains Siano.

His team also began using lasers for treating frescoes and one of the most interesting examples is the 2009–10 restoration of the Santa Tecla catacombs beneath Rome. The early Christian paintings in this damp underground environment were covered with a hard-to-remove calcareous crust. Siano says this was ‘a conservation problem that was absolutely insoluble with the traditional approaches’. The laser cleaning uncovered the earliest known images of the apostles Paul, Peter, John and Andrew on the ceiling of a fourth century AD tomb.
Siano is now turning his laser to removing old varnishes and restorations from easel paintings. He is still using a Nd:YAG laser, but passing the 1064nm beam through a nonlinear optical crystal to produce a visible green second harmonic wavelength of 532nm. At this lower wavelength, the laser cleans by causing photomechanical surface expansion. Siano used this method to restore a female portrait from the studio of the Italian Futurist artist Giacomo Balla (1871–1958). While not a standard option for paintings, Siano estimates that laser systems are now regularly employed for sculptures, bronzes and frescoes by more than 400 European conservation institutions.
A soft touch with nanoscience
Another major innovation in the last decade is the use of colloid science and nanotechnology in conservation. The starting point was a problem presented to another University of Florence scientist, colloid chemist Piero Baglioni, in the mid-1980s: how to remove beeswax spots from Renaissance frescoes in the Brancacci Chapel in Florence. Traditional solvent cleaning was not working because of surface porosity, as Baglioni recalls. ‘If you solubilise the wax, [it] penetrates even more deeply inside the walls.’
Baglioni came up with a microemulsion: a clear mixture of organic solvent and water, stabilised with a surfactant that sits at the interface between the water and organic phases. The organic phase in the microemulsion forms small 5–15nm sized droplets, creating a huge surface area to act like a detergent, dissolving in this case wax. The dissolved wax is prevented from spreading, because the organic phase is held within the microemulsion, The organic solvent is less than 2% of the microemulsion, which decreases the overall toxicity dramatically.
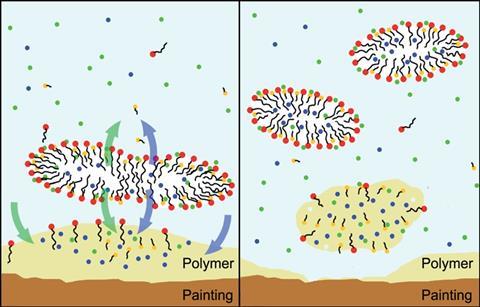
Baglioni was again called upon for an answer when the removal of polymer coatings applied to frescoes in the 1960s became a priority. Ageing rendered the polymers insoluble to most organic solvents because of oxidation and cross-linking reactions, but Baglioni created a series of microemulsions that did the trick. In Mexico, polymer coatings were applied to frescoes as late as the 1990s; within ten years it was clear this was a mistake. In 2008, Baglioni started to work with the Mexican National Archaeological Institute to come up with a removal treatment for murals at the Mayan site Mayapan.
The system they came up with consisted of a number of solvents including pentanol, propylene carbonate and ethyl acetate. It had a structure known as a ‘swollen micelle’, which is on the borderline between a classical microemulsion and a micellar solution, where surfactant molecules aggregate into micelles – nano-sized spherical clusters. In a swollen micelle, a number of the solvent molecules are found in both the water phase and organic droplets and this proved to be the best and most versatile system, able to remove a range of polymers. Baglioni suggests the solvent molecules dissolved in water are first able to diffuse into the polymer coating to swell and break down the polymer, followed by more solvent diffusing from the nanodroplets into the water allowing the continued breakdown and detachment of the polymer.
Rebuilding work
Baglioni has also pioneered the use of nanoparticles for repairing deteriorating frescoes. Artists generally painted directly onto wet calcium hydroxide plaster, which reacts with atmospheric carbon dioxide to form calcium carbonate (calcite). Over centuries, pollution and humidity causes the carbonate layer to break down and sulfate, nitrate and chloride salts within the walls recrystallize, leading to deterioration of the painted surface. Baglioni was sure that nanoparticles would improve on conventional conservation methods. ‘We thought that if we put the calcium hydroxide back into the paintings, we can reform the carbonate pieces in situ.’ His treatment injects calcium hydroxide nanoparticles dispersed in alcohol and their small size, just 10–100nm, allows them to penetrate several centimetres into the frescoes and slowly reform the depleted calcite.
We can reform the carbonate pieces in situ
Piero Baglioni
Since developing the nanoparticles in the late 1990s, Baglioni realised they would also work for paper and canvas conservation. The breakdown of cellulose fibres in paper and fabric occurs through acid-catalysed hydrolysis and leads to the characteristic yellowing and embrittlement. Using hydroxide nanoparticles, paper and canvas is deacidified with the excess particles quickly converted into carbonate, providing a protective neutral pH.
Baglioni explains size is the crucial criteria in using nanoparticles. ‘Depending on how fast or slow you want the formation of calcite to be, you play with the size of the particle.’ Smaller particles will have faster reaction times. For the consolidation of frescoes, larger 100nm particles might be selected. Their size balances the ability to penetrate into a fresco with a reaction rate slow enough to produce large mechanically strong calcite crystals. Whereas for the conservation of paper, where a high pH can be damaging, smaller 50nm particles are preferable so that excess particles form calcite within hours.
The metabolic power of bacteria

An unusual approach to cleaning came from the Polytechnic University of Valencia, Spain. We are familiar with enzymes in washing powders but Pilar Bosch’s team has taken things a step further, using bacteria to remove inorganic crusts and animal glues from frescoes. Because bacteria can produce a whole host of enzymes they can deal with complex cleaning problems, metabolising organic and inorganic matter into hydrogen sulfide, molecular nitrogen or carbon dioxide. Over the last 10 years, the method has been successfully used at several important sites, including the Camposanto in Pisa, Italy, a 13th century burial ground supposedly constructed around soil brought back from Jerusalem during the Crusades. It was decorated with extensive frescoes that have been badly damaged.
Biologist Bosch worked with a team of microbiologists, chemists and conservators to develop the treatment. She says they started in the microbiology lab: ‘We have to find the micro-organisms that are able to metabolise the substances we want to clean without damaging the artwork.’ All bacteria tested are harmless and do not form spores. Over several projects, Bosch tested different strains of the nitrate-reducing bacteria Pseudomonas stutzeri, commonly found in soil and water. The strains chosen were able to remove nitrate crusts and animal glues when applied with agar gel and left for between several hours and several days.
Bosch says that there are no signs of any problems with the frescoes treated ten years ago and, although there is sometimes a knee-jerk fear of using bacteria, interest in the method is growing, with several companies investigating making commercial kits. She is looking at other applications, for example removing glue from paper.
An acrylic time-bomb
The introduction of organic dyes in the 19th century, followed by paints based on acrylic polymers in the 20th has set a conservation time-bomb ticking. The survival of vivid colours in Renaissance art is largely due to the chemical stability of the inorganic pigments used, but this is not the case with modern acrylic paints. While the scale of the problem is not yet clear, it is likely that within several hundred years synthetic dyes will have faded and plastic binders depolymerised, becoming powdery. ‘Being provocative, we can say that if you looked at the Museum of Modern Art in New York in 100 years it will be an empty museum, because what’s inside will be completely degraded,’ Baglioni says.
We need to ensure that what we do stands the test of time
Bronwyn Ormsby
Baglioni is starting to bring his knowledge of microemulsions to bear on this unsolved problem. He says that while the chemistry is complex, he has some ideas of how to reverse the chemical degradation and hopes in the future to develop cleaning gels able to reformulate original compositions. The Tate Gallery also has a significant stake in the conservation of modern acrylic paints and Ormsby is involved in ongoing research on their conservation.
A number of the innovative methods described have become standard conservation practices. Laser cleaning is well established and Baglioni’s nanoparticles are commercially available, but as the name suggests, conservation is a conservative field and the adoption of new methods is rarely fast. Siano describes a ‘natural inertia’ in conservators which he thinks is responsible for holding back the early uptake of laser ablation methods, but Baglioni concedes that ‘in some cases it’s easy’, where there is an urgent need, such as for polymer removal.
In the long term, we may be grateful for a measured, scientific mindset in conservation. ‘Works of art are unique and valuable, so we need to ensure that what we do – whether it’s removing something or adding something – is guided by conservation ethics and stands the test of time,’ Ormsby says. With an ethical and innovative approach, there is hope that modern acrylic art will last as long as the Sistine Chapel frescoes.
Rachel Brazil is a science writer based in London, UK
Correction: This article was updated on 25 June 2014 as the affiliation of Salvatore Siano was wrong. It is now correct.











No comments yet