James Mitchell Crow finds that the outlook for renewables-powered electrochemical ammonia production is beginning to brighten
Karthish Manthiram dreams of an electrified future. The MIT chemical engineer is among a growing band of researchers exploring ways in which renewables-powered electrochemistry could replace fossil fuels as the foundation of the chemical industry.
‘The overall vision is to think about a future in which chemical manufacturing begins with just carbon dioxide, water and nitrogen,’ Manthiram says. ‘If we start with these three feedstocks, and we have renewable electricity, then we can envision using carbon dioxide as a carbon atom source, using water for oxygens and hydrogens, and dinitrogen as a source of nitrogen, and start to stitch together relatively complex organic molecules,’ he says.
In that future world we are dreaming of, we could make ammonia from just air, water and renewable electricity
High on the list of molecules that electrochemistry researchers want to make this way is ammonia. A key ingredient in synthetic fertilizer, approximately 175 million tonnes of ammonia is produced annually. But the vast quantities in which it is made, and the energy-intensive nature of its production, means ammonia plants consume approximately 2% of global energy production, and produce a similar proportion of global carbon dioxide emissions.
With the rapidly increasing availability of renewable electricity, researchers can see an alternative pathway to ammonia. ‘In that future world we are dreaming of, we could make ammonia from just air, water and renewable electricity,’ Manthiram says. This year, Manthiram’s lab joined the handful around the world to have definitively shown that, when mediated by a lithium electrocatalyst, ammonia could be made this way.
In a young research field in which early claims of electrochemical ammonia production have often not stood up to scrutiny, the handful of robust results involving lithium-mediated ammonia production could form the solid foundation from which a clean ammonia future begins to be built.
Splitting headache
The great challenge of ammonia synthesis, electrochemical or otherwise, is the requirement to take dinitrogen from the air, and break it in two. The triple bond holding the two nitrogen atoms together is one of the strongest known.
For the past century, ammonia plants have overcome dinitrogen’s reticence to react via a two-step process. In the first step, the steam-methane reforming reaction, water and methane are heated over a catalyst that encourages atomic partner swapping, producing a stream of carbon dioxide and hydrogen. The hydrogen is then reacted with nitrogen over an iron catalyst in the Haber–Bosch process, named after German chemists Fritz Haber who invented the reaction, and Carl Bosch who refined it for industry.
A large part of our inequity in access to food in the world is traced back to the fact ammonia production is centralised
What Haber discovered was a way around the problem that, at the high temperatures needed to split dinitrogen, the equilibrium for ammonia formation lies far to the left, says Ib Chorkendorff, a catalysis researcher at Technical University of Denmark, and director of the Villum Center for the science of sustainable fuels and chemicals. ‘Haber’s big invention was to realise that you won’t get any ammonia unless you pressurise it,’ Chorkendorff says. ‘If you take four moles of gas and turn it into two, pressure helps a lot.’ Haber Bosch plants today typically run at pressures in excess of 200 atmospheres.
The stoichiometric carbon dioxide the reaction produces, as well as the carbon emissions involved in generating the high temperatures and pressures required, is one compelling reason to look for an alternative approach. ‘But even if carbon dioxide were not an infrared trapping gas that leads to global warming, we’d still have a problem with the process,’ Manthiram says. ‘The really high temperatures and pressures mean it is just not possible to do this reaction in a distributed fashion.’
The cost of building high pressure reactors tips the economic equilibrium heavily toward the construction of very large, centralised ammonia plants. ‘A large part of our inequity in access to food in the world is traced back to the fact ammonia production is centralised,’ Manthiram says. ‘In sub-Saharan Africa, the distribution infrastructure is so poor that the fertiliser ends up costing two to three times the international average. This limits its use, which leads to low crop yields, hunger, lower workforce productivity, lower earnings, which further limits the ability to afford the fertiliser. This is an enormous problem to solve.’
Ammonia made from air, water and renewable electricity, at ambient temperatures and pressures, could be made wherever it is needed.
Feeling renewables’ pull
Researchers have tried for decades to find greener alternatives to the Haber–Bosch reaction. The difference this time is the rapidly changing energy market, says Doug MacFarlane, a chemist at Monash University who heads the Australian Research Council Centre of Excellence for Electromaterials Science. ‘In the last 10 years or so, increasing amounts of increasingly inexpensive renewable energy has become available,’ MacFarlane says. Since 2010, the price of solar energy has fallen 80%, and wind energy by 50%, with the prospect of further falls. ‘That has put the focus on electrochemical technologies very broadly, using electrons as the ultimate reductant,’ he says.

MacFarlane’s lab – like many electrochemical synthesis labs – initially focused on renewables-driven water-splitting chemistry, to produce a stream of clean hydrogen that could be used as a feedstock or fuel. The second wave of research was focused on carbon dioxide reduction, to turn carbon emissions into useful products, he says. ‘Growing out of that, about six years ago we began to realise our research had a third prospect to it, which was nitrogen reduction,’ MacFarlane says. ‘It’s the same ideas, same laboratory kit, but a different substrate and target product.’ Rather than hydrogen, the cell would produce a stream of ammonia.
Ammonia production could go far beyond fertiliser use. Falling electricity prices driven by renewables has already prompted a rethink of the traditional steam reforming reaction (see box Electric reformation below). Countries such as Australia, which have solar and wind resources far beyond their own needs, are eyeing electrochemically synthesised ammonia not only for cleaner fertiliser production, but as a green fuel that could enable the export of renewable energy.
In May 2020, MacFarlane and his colleagues published a roadmap laying out the potential route to a green ammonia economy. The first-generation concept, the idea of combining conventional ammonia production with carbon capture, is already being superseded by a second-generation approach, in which the hydrogen for Haber–Bosch comes from renewables-powered electrochemical water splitting, rather than from methane-driven steam reforming.
After decades of research, electrochemical water splitting has reached the point that industrial-scale water splitters are becoming available, from the likes of ITM-Power in the UK and Siemens in Germany. Siemens has already tested the concept of combining water splitting and Haber–Bosch at the Siemens Green Ammonia Demonstrator in Oxfordshire. In the Pilbara region of Western Australia – which receives some of the highest annual solar energy of anywhere on the planet – Yara Fertilisers is also preparing to pilot the switch. This plant currently produces 5% of the world’s ammonia.
The main barrier to wide uptake of second generation ammonia production is no longer the cost of renewable electricity, says MacFarlane. ‘The barrier is the capital cost of the electrolysis cells,’ he says. The best-performing electrodes for water splitting use precious metals.
Electric reformation
The first step in conventional ammonia synthesis is to generate the hydrogen that will be combined with dinitrogen to make ammonia. Today, that hydrogen comes from methane, via the steam reforming reaction, driven by an array of gas burners surrounding the catalyst-coated tubes in which the reaction takes place.
The amount of catalyst can be reduced by an order of magnitude
Steam reforming is so endothermic, it generates a significant temperature gradient within the tube, which can lead to the build-up of carbon on the catalyst surface and the blockage or even rupture of the tube.
In 2019, Chorkendorff showed the reaction could be driven much more effectively by ohmic heating. Passing an electric current through the steel steam reformer tube generates heat in situ, due to electrical resistivity. ‘We get a very close contact between the heating source and the catalyst, which eliminates all the heat transport problems and we have no temperature gradient at all,’ Chorkendorff says.
Aside from avoiding carbon deposition, ohmic heating offers many advantages, Chorkendorff adds. To start up a steam reforming plant usually takes roughly a week, but the ohmic-heated system takes about two minutes. ‘The amount of catalyst can be reduced by an order of magnitude, and the size of the plant by two orders of magnitude,’ he says. Previously, the cost of electricity made ohmic heating prohibitively expensive – but with renewables, that is no longer the case, Chorkendorff says. The company Chorkendorff partnered with to carry out the research is already working on a pilot plant, and has plans for ohmic heating far beyond steam reforming, he says.
Cutting costs
Finding lower cost alternatives to platinum and iridium electrodes has been a long-term target of Chorkendorf’s lab. In 2005 the team showed molybdenum sulfide makes a strong electrocatalyst for the hydrogen evolution reaction. The catalytically active part of MoS2 has turned out to be along the catalyst’s edges, rather than its flat surface, prompting a lot of research into developing nanostructured or layered MoS2 materials that maximise the proportion of edges.
We know an OER catalyst has to be an oxide and it has to be stable to extreme pH
‘We have looked a lot at hydrogen evolution, but we consider that solved – there are some very good catalysts,’ Chorkendorff says. ‘The real problem is the energy lost due to oxygen evolution,’ he argues. Water-splitting cells produce hydrogen at the cathode and oxygen at the anode (see box Meanwhile, at the anode below). Low efficiency on the oxygen evolution reaction (OER) side of the water splitter makes the whole process more energy-intensive and costlier to run. ‘Today they run at an efficiency of maximum 70%, but normally not that high.’
Despite years of work, the OER catalyst breakthrough is yet to come. ‘There is nothing saying we can’t find a material, but we know it is getting tight – we know it has to be an oxide, it has to be stable either in strong acid or strong alkaline solution,’ Chorkendorff says. Electrolysers are run at one extreme or the other of the pH scale to maximise efficiency, but the most active OER catalysts are not stable enough under these conditions for long term practical use.
One concept, introduced by Daniel Nocera, then at Massachusetts Institute of Technology in the US, is self-healing OER catalysts, which form in-situ and continually regenerate from metal ions in the electrolyte. Nocera’s electrocatalysts operated in neutral conditions. In 2019, MacFarlane and his colleagues demonstrated a self-healing, in situ generated mixed metal oxide OER catalyst for strongly acidic conditions.
Meanwhile, at the anode
For most electrochemical processes – making hydrogen or ammonia, or reducing carbon dioxide – all eyes are on the cathode. The companion reaction taking place at the anode is an oxidative process that typically generates oxygen, an innocuous by-product to the main event.
‘Many people consider the oxygen as just a waste product,’ says Chorkendorff. But there are potentially valuable uses for the gas. A stream of pure oxygen is key to the concept of oxyfuel powerplants, which sidestep carbon capture issues by burning fuel in oxygen rather than air. ‘If you burn biomass in a powerplant with that oxygen, you get a much higher grade of carbon dioxide,’ Chorkendorff says.
Manthiram also sees value in the anode reaction. ‘We have been working on methods by which you can use that oxygen for chemical synthesis, instead of trying to combine two oxygen atoms to make dioxygen which is kinetically difficult to do.’ From prior work, the team knew that manganese oxide is a catalyst that oxidises water, generating a manganese oxo species in the process. ‘What if we could intercept that using an olefin, to make an epoxide?’ Manthiram asks. ‘It was purely conjecture, but somehow it worked.’ As well as producing epoxides, the team has shown it can insert oxygen into a carbon–carbon bond to make lactones. ‘It could make the water electrolyser more economically feasible,’ Manthiram says. ‘There’s no way to make all the world’s hydrogen this way – we have far too great a need for hydrogen – but we could make some hydrogen that is really cheap while co-creating lactones that go into high-volume biodegradable plastics, or high-volume epoxides to make ethylene glycol.’
Unreliable sources
With water splitting still to be perfected, and with its continued reliance on the Haber–Bosch reaction, the hope is that second-generation green ammonia production is a stepping stone toward the ultimate goal of producing ammonia directly by electrochemical means. Rather than produce dihydrogen gas, hydrogen and nitrogen atoms could be combined in situ at the cathode to make ammonia.
The main challenge is finding ways to favour ammonia production over dihydrogen formation, Manthiram explains. ‘The reservoir of electrons at the cathode could react with two protons in solution to make hydrogen; or instead react with nitrogen to break the triple bond, and also recruit six protons, to make two ammonia molecules,’ Manthiram says. ‘Virtually any catalyst will tend to make more hydrogen than ammonia.’
Over the past five years or so, a stream of papers appeared that seemed to show catalysts that could produce some ammonia in the predominantly hydrogen gas stream produced at the cathode. Meanwhile, MacFarlane’s lab was testing the idea that a good way to supress hydrogen production would be to eliminate one major source of protons – the electrolyte. Switching to an aprotic ionic liquid electrolyte, and then just adding the controlled amount of water, acid or base, the team showed they could favour ammonia production.
Virtually any catalyst will tend to make more hydrogen than ammonia
‘The aprotic work is quite agnostic about the catalyst, so our approach was always to apply it to the best catalyst that we could find,’ MacFarlane says. ‘Whenever a paper on a new catalyst was published, we would quickly try to repeat the literature work, then apply it in our aprotic work. In some frustration, in some cases after talking to the authors involved, we have never managed to get a catalyst from the literature to work well. We ended up becoming experts in what doesn’t work.’
Although transforming nitrogen into ammonia is very difficult, it is all too easy to produce traces of ammonia from other nitrogen sources. The long list of possible sources of adventitious nitrogen include the electrode, or traces of NO or NO2 in the nitrogen gas supply. Strip out or account for these sources, and the amount of nitrogen being converted to ammonia by most catalyst systems is so low it is of no practical interest, MacFarlane says.
‘[MacFarlane] has been a huge proponent of figuring out where the ammonia is actually coming from and whether the catalyst is really doing the work,’ says Lauren Greenlee from the University of Arkansas in the US, who was one of the first to work on electrocatalytic ammonia production, and one of the first to highlight that adventitious ammonia was a major concern. ‘Directly converting dinitrogen to two ammonia molecules on the surface of the catalyst is just a very hard problem, and I don’t know that we are that close to solving it.’
For now, Greenlee has stepped back from ammonia production experiments to carry out more fundamental research into the problem. ‘We got interested in the chemistry of electrolyte, and working to understand the catalyst surface chemistry and how it changes with these electrolytes,’ Greenlee says. ‘I don’t make any promises about making ammonia, but hopefully we will learn a lot about the catalyst and contribute to that field.’
Looking to lithium
As well as Greenlee and MacFarlane, Chorkendorff has set out the rigorous control experiments, including using radiolabelled dinitrogen, needed to show that the ammonia produced is indeed coming from dinitrogen and not an alternative nitrogen source. ‘We published a method, because I am sick and tired of looking at all those papers claiming they are making ammonia, because they are not making any,’ Chorkendorff says. ‘I only know one system that is making ammonia for sure, and that is the lithium-mediated route.’
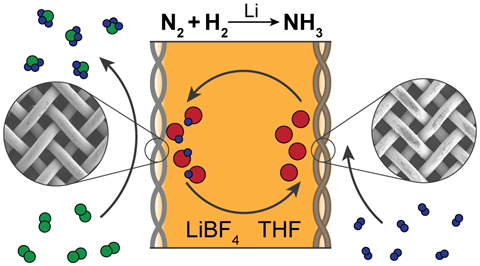
Lithium is the only metal that can react spontaneously with dinitrogen to form a metal nitride – breaking the dinitrogen triple bond in the process. Lithium’s capability to generate ammonia was demonstrated by Japanese researchers in the mid-1990s – the only protocol Chorkendorff was able to reproduce.
The lithium-mediated approach to ammonia is currently the only one that without a shadow of a doubt works, MacFarlane agrees. Lithium’s downside is that it involves a heavy energy efficiency penalty, requiring an overpotential of more than 3V. ‘This is a lot of energy that will be wasted, lost as heat, which is why it hasn’t been picked up and developed previously,’ MacFarlane says.
But what the protocol does have going for it is the fact it definitely works – and it offers plenty of directions for future research. ‘The lithium-mediated approach is our main approach at the moment,’ MacFarlane says. ‘One obvious way to solve the energy problem is to look at other metals, and their metal nitrides, that would carry out this process at a lower energy cost, a less negative energy potential,’ he says.
In Chorkendorff’s lab, the focus is on understanding what makes lithium effective in the first place. ‘We are first of all figuring out what actually sits on that surface to carry out that process. We are using the synchrotron at Stanford, trying to build a model for the process.’
A couple of years ago, the rates were so low we didn’t even know if we were making ammonia
Manthiram, meanwhile, has been looking at ways to tackle the problem of efficiently getting gases to react in a liquid-based electrochemical cell. ‘We demonstrated how one could use a gas diffusion electrode as a means of introducing nitrogen gas much more effectively into the reactor, overcome transport limitations, and thereby make ammonia at much faster rates and higher selectivity for making ammonia versus hydrogen,’ he says. The team achieved a record ammonia yield.
So far, the gas diffusion reactor only operates over a few minutes, and at an extreme overpotential. ‘They’re sort of forcing it – but despite the drawbacks, I like that paper very much,’ Chorkendorff says. ‘It shows the way forward and the science that is needed.’
The team – and the field – has its work cut out to turn the process into a practical solution for fertilising crops, or for storing and transporting renewable energy, Manthiram says. ‘But when we think back to where we were in this field a couple of years ago, the rates were so low we didn’t even know if we were making ammonia. We are finally at the point, I think our paper shows, that rates are so high you actually don’t need to do a control experiment with labelled nitrogen to demonstrate that that ammonia is real. There is just no way there could be adventitious ammonia at those concentrations.’ Although he hastens to add that the team still does the control experiments.
For now, the team is putting up with lithium’s inherent overpotential problem. ‘It is the toll one has to pay to go down this road, and we have come to terms with paying that price, because there is no other system that can operate even in the vicinity of the rates we have achieved with lithium,’ Manthiram says. ‘I would be surprised if other methods did not emerge in the next decade that did not pay that penalty,’ he adds. ‘But this is where the field is right now.’
James Mitchell Crow is a science writer based in Melbourne, Australia












2 readers' comments