Green ammonia promises a more sustainable future. Jamie Durrani talks to the researchers aiming to revolutionise the production of crucial fertiliser
In the early 20th century the newly developed Haber–Bosch process transformed food production. Reliable access to ammonia-based fertilisers was a huge boost to agriculture and helped to sustain the planet’s rapidly growing population.
‘When Haber and Bosch discovered how to make ammonia artificially, that triggered a whole chemical revolution – it was the very first chemical process used at that scale,’ explains Laura Torrente-Murciano, an expert in reaction engineering and catalysis based at the University of Cambridge, UK. ‘It was created to be used as explosives, mainly, but indirectly, it is used today for fertilisers. And therefore, it sustains the world … around 50% of the world population are fed by ammonia made in chemical reactors.’
But the Haber–Bosch process hasn’t changed all that much since its discovery more than 100 years ago. The process uses iron or ruthenium catalysts to react hydrogen and nitrogen together under extreme conditions. Temperatures can reach 600°C, with pressures raised to over 200 times that of the Earth’s atmosphere.
Ammonia is going to play a key role in the decarbonisation of the society
Today, giant Haber–Bosch reactors produce around 180 million tonnes of ammonia each year, using up more than 1% of the total energy produced worldwide. The process is also intertwined with fossil fuel extraction – the hydrogen feedstocks are made by steam reforming coal and natural gas – and is directly responsible for 450 million tonnes of carbon dioxide emissions annually.
With the urgent need to cut society’s carbon emissions, Haber–Bosch is long overdue a makeover. Many researchers now believe that sustainably produced ‘green’ ammonia would not only reduce the environmental impact of fertilisers, but could also be used as a clean energy carrier.
‘If you think about the molecule itself, it is not that different to the methane molecule. So ammonia has one nitrogen, three hydrogens; methane, one carbon, four hydrogens. And that means that they store and release energy in a very similar way,’ says Torrente-Murciano. ‘So I think ammonia is going to play a key role in the decarbonisation of the society because it can be used in a very similar way that we use methane today.’
Engineering issues
Transforming ammonia production is likely to move in two stages. The first involves adapting current production methods so that green hydrogen can be used as a feedstock, with renewable electricity used to power the plants. Further into the future, new methods that rely on completely different chemistry could come online.

While adapting existing facilities to use green hydrogen might sound straightforward, it is not quite so simple because current Haber–Bosch plants are entirely planned around the steam reforming process.
‘[The current Haber–Bosch process] it is designed around using fossil fuels as the feedstock for hydrogen and also as a source of energy to run the whole process. The problem is, when you’re trying to make green ammonia, that whole integration disappears,’ says Torrente-Murciano. ‘If you produce hydrogen in a different way, you need to think about ways of running your compressors in the ammonia loop, how you’re going to heat your reactors, how you’re going to start up your systems and things like that – that requires a completely new optimisation of the process.’
The very high pressures associated with Haber–Bosch help to maximise the amount of nitrogen and hydrgeon that is converted into ammonia in a single pass, without having to be fed back into the reactor. In current facilities, the compression systems are based on steam that is a byproduct of the reaction that makes the hydrogen feedstock from fossil hydrocarbons. But if the process is to be based on green hydrogen, it would make sense to use much more energy-efficient electric compressors.
Producing green hydrogen with renewable electricity also brings additional challenges – chief among them being the problem of intermittency. Haber–Bosch facilities are huge sites that are designed to run 24 hours a day, seven days a week, requiring a steady supply of hydrogen to feed the reactors.
One solution is to build up a hydrogen buffer – a large store of hydrogen that is bolstered when the sun is out, or the wind is blowing, and which the ammonia plant can draw upon continuously. But this would only help to address short-term energy fluctuations and brings significant extra costs.
‘The other solution is the development of novel ammonia synthesis technologies that are able to shut down production and go back again depending on whether it is sunny or not,’ says Torrente-Murciano. ‘And that’s a huge change of way of thinking about the chemical industry … But if we are able to do that, it will be transformative.’
Torrente-Murciano and her colleagues have recently demonstrated a system in which ammonia is separated by absorption rather than condensation. This system operates at pressures as low as 30 bar and the production and separation of ammonia can take place in a single vessel, resulting in a much more flexible process.
‘It is important to note that this is a highly exothermic reaction, thus heat management is a challenge – in particular if we are operating the system in a dynamic manner,’ she adds. ‘We need to be prepared for distributed manufacturing of ammonia to be aligned with the distributed nature of the renewable energy – with potential plants orders of magnitude smaller than the current ones, where dynamic operation would be more manageable from a heat point of view.’
Lessons from nature
Beyond just modifying current Haber–Bosch processes, many researchers are attempting to design fundamentally different ways of making ammonia. But there’s a reason why we’re still using a century-old process. Nitrogen, which makes up almost 80% of the air around us, is an exceptionally stable molecule. Nitrogen’s triple bond is the strongest homodiatomic bond known in chemistry, and breaking it is no simple task.
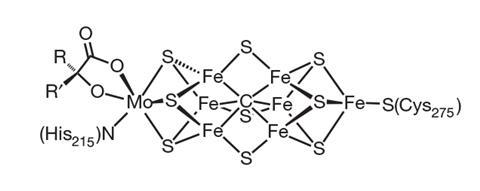
In nature the nitrogenase family of enzymes does the job. These enzymes are found in microbes that fix nitrogen into bioavailable ammonia. ‘You might speculate that in the evolution of the planet, plant life really couldn’t have gotten very far if it hadn’t been for nitrogenase producing fixed nitrogen – because the nitrogen content of the current biosphere all comes from that enzyme,’ says Doug MacFarlane, whose group at Monash University in Melbourne, Australia, is developing electrochemical means to produce ammonia.
MacFarlane explains that many researchers seeking to develop new nitrogen-fixing processes are keen to take lessons from how nature does it. There are three main classes of nitrogenase and all feature a core containing iron and sulfur atoms, but they are distinguished from each other by the presence of molybdenum, vanadium or just iron.
‘For people doing homogeneous or even heterogeneous catalysis, these look almost like pieces of a heterogeneous surface or they look like molecules you could imagine to synthesise,’ says Serena DeBeer, whose lab at the Max Planck Institute for Chemical Energy Conversion in Mülheim, Germany, uses an array of x-ray and spectroscopic techniques to probe the nitrogenase enzymes’ activity. ‘And so there’s a lot of interest in understanding exactly how does this heterometal tune the reactivity? How does it enable a different kind of reactivity?’
‘That said, all of these proteins are part of large multicomponent protein systems, and they’re also controlling the delivery of electrons and protons,’ she adds. ‘And so much of what we’re trying to understand is at each given step: where are the electrons going? Where are the protons going? And how does that vary in these different forms of the enzyme?’
‘They have dramatic differences in reactivity, that’s clear. They have intrinsically different catalytic active site, that’s clear. But it seems to hold a lot of intrinsic chemistry lessons designed by biology that we’re trying to tease out.’
Despite decades of research into nitrogenase’s activity, exactly how nitrogenases produce ammonia is still not fully understood. The main reason for this is that nitrogenase itself is a uniquely complicated enzyme.
‘Every step of the way, the crystallographers have been challenged, the spectroscopy has been challenged – part of it comes from the fact that this is a challenging protein to work with,’ says DeBeer. ‘But part of it is just even studying the nature of such a complex cofactor – it’s a cofactor with seven irons, one molybdenum, all open-shell, all spin-coupled … There isn’t a more complex cofactor anywhere in biology.’
Even theoretical scientists struggle to get to grips with nitrogenase, and DeBeer notes that density functional theory can’t fully describe the enzyme’s complicated nature. ‘For almost everything we do, it has a level of complexity in terms of the structure, the magnetism, the mechanism that goes beyond anything we have a parallel to.’
Small molecule catalysts
While there are many question marks surrounding nitrogenase’s action, the enzyme does offer pointers for those chemists trying to create alternative routes to ammonia. Key aspects of the enzyme’s activity are in the way it first binds the dinitrogen molecule and then delivers electrons and protons in a controlled fashion to avoid just making hydrogen. Avoiding this competing hydrogen-evolution reaction is a major challenge for researchers working across many different approaches to nitrogen reduction – from those developing small molecule catalysts to those working on electrochemical approaches.
In 2003, Dmitry Yandulov and Richard Schrock demonstrated the first example of a homogenous catalytic nitrogen reduction using a transition metal complex. This first molybdenum catalyst was partially inspired by the iron–molybdenum cofactor present in some nitrogenases, and further molybdenum catalysts like those developed by Yoshiaki Nishibayashi at the University of Tokyo, Japan, have since achieved nitrogen reduction rates approaching those of nitrogenases.
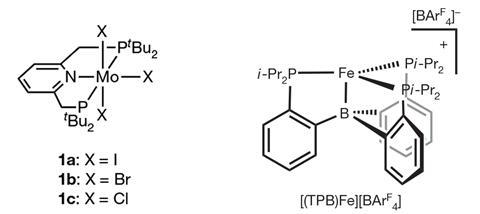
In light of growing evidence that nitrogenase’s iron atoms might actually be more important for binding nitrogen molecules, the group of Jonas Peters at the California Institute of Technology developed the first iron-based catalysts that are capable of converting nitrogen into ammonia. Nishibayashi’s group also developed iron catalysts using similar pincer ligands to those used with their earlier molybdenum systems. Catalysts based on other metals have also been demonstrated, but so far only the molybdenum ones work at room temperature, with others requiring cryogenic temperatures.
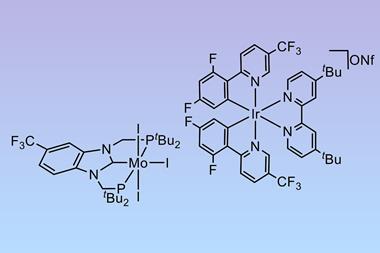
Independently of each other, Peters’ and Nishibayashi’s groups have very recently developed similar photocatalysts that are capable of reducing nitrogen to ammonia.
‘The Haber–Bosch process is a reaction driven by the energy of the reactants. For example, in the Haber–Bosch process, hydrogen gas is the energy source,’ says Nishibayashi. ‘In contrast to conventional reaction systems including the Haber–Bosch process, ammonia synthesis using visible light is an ammonia synthesis method that uses energy from external sources. We believe that this achievement provides an opportunity to develop ammonia synthesis reactions using renewable energy.’
The systems developed by both Peters and Nishibayashi’s groups use molybendum-based nitrogen reduction catalysts featuring pincer ligands in combination with iridium photoredox catalysts. Both systems require sacrificial proton donors, and Nishibayashi notes that this is one area that his team hope to improve.
‘Our future goal is to develop an ammonia synthesis reaction using visible light energy with water instead of dihydroacridine as a sacrificial reducing agent and hydrogen source,’ he says. ‘We will also try to dramatically improve the quantum yield.’
Wiring the fixing
One of the most promising approaches to artificial nitrogen reduction involves electrocatalysis. Some researchers have even used nitrogenase in conjunction with electrodes to carry out nitrogen reduction. However, these systems are at a very early stage of development and are currently challenged by low rates, instability and poor efficiency.
Many other systems have been analysed using different types of electrodes. However, this emerging field has had problems with reproducibility. One of the main issues for researchers investigating new systems is that until a catalyst reaches a certain level of activity it is very difficult to tell whether any ammonia is produced electrocatalytically, or if it has derived from nitrogen-containing trace impurities that are present in the air and in solvents.
‘There’s a real problem in really proving [when] anybody’s done anything particularly impressive in this field,’ says MacFarlane. ‘The problem is that the yield rate and the faradaic efficiencies are so low, that it’s quite possible that [any ammonia] is coming from contaminants.’
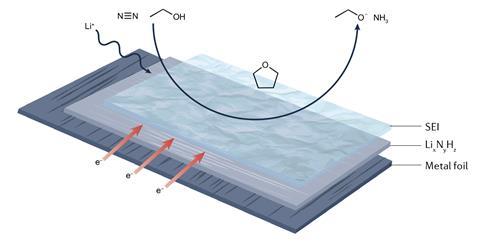
To date, the only electrocatalytic method for which there is irrefutable evidence of successful nitrogen reduction involves splitting the dinitrogen bond using an electrode plated with metallic lithium. These systems generally use an electrolyte formed of an organic solvent containing a lithium salt and proton carrier – often ethanol.
‘Essentially the key to enabling selective nitrogen reduction rather than binding protons and making molecular hydrogen is to constrain the access to either electrons or protons,’ says electrochemist Ifan Stephens from Imperial College London, UK. ‘That’s what happens in nitrogenase, and that’s what happens in molecular catalysts. And that’s what we think is happening in the system we’re looking at, which is the lithium-mediated electrochemical system.’
‘The field’s come a really, really long way,’ notes Olivia Westhead, who works alongside Stephens at Imperial. ‘But there’s still a lot more fundamental understanding that we need and a lot more optimisation to make it more scalable and industrially relevant.’
She notes that concentrations of salts, ethanol and water in the electrolyte can dramatically alter the efficiency of the lithium-mediated nitrogen reduction system. ‘It’s not really great industrially if your system is so dependent on this many factors,’ she says. ‘That’s something that needs to be looked into – making it more resilient to the small changes.’
While early examples of lithium-mediated nitrogen reduction suffered from low efficiency and poor stability, recent studies appear to have dramatically improved these metrics. A major factor in the performance of lithium systems is the solid electrolyte interphase layer (SEI), which forms on the surface of the negative electrodes during operation. The SEI improves the stability of the electrode potential and also helps to constrain the number of protons that reach the electrode surface – helping to prevent hydrogen evolution.
‘There’s been quite a few different ways people have looked at addressing the [stability] problem,’ says Westhead. ‘In our group, we saw that if you just increase the salt concentration, the negative electrode potential stabilises and that’s because you improve your SEI – you make it much more inorganic and much more stable.’
‘Other groups have done it differently. [Researchers at] the Technical University of Denmark found that if you add a bit of oxygen, it stabilises your SEI layer as well – so then you could run it for ages,’ she adds. ‘They also develop a pulsing protocol where if you pulse your current the negative electrode potential stays stable for much longer.’
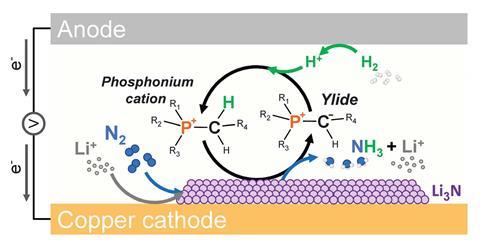
Some of the most dramatic improvements have come from changes to the proton carrier and electrolytic salt. In 2021 MacFarlane’s group showed how the use of a phosphonium proton shuttle, not only helps to deliver protons in a controlled manner, but also increased the ionic conductivity of the system.
‘The other thing that was pretty important was that the proton donation from the phosphonium and the reprotonation was all reversible – there is no sense in which this proton carrier can be used up,’ says MacFarlane.
Magic fluoride
At the same time, MacFarlane’s group was investigating the role of the lithium salts. He explains that research on lithium batteries has shown how important the role of the salt can be, but previously only a small number had been tested in ammonia reduction. In particular, his team was interested in finding out if fluorinated imide-based lithium salts might enhance the performance of the ammonia-producing cell.
‘It’s known in the field that those salts are very much more soluble in these ether solvents – in our case THF. And this allowed us to explore a range of concentrations vastly higher than had been the classical work, which tends to be one molar or below, sometimes as low as 0.1 or 0.2 moles per litre – which is not a lot of conductivity,’ he says. ‘Suddenly in that work, we went all the way up to about four moles per litre, which is a lot of salt.’
By using this much more concentrated electrolyte, conductivity and current densities in the cells were suddenly much higher. This drove up the activity of the cell and achieved almost 100% current to ammonia efficiency.
‘We realised that we were getting to the point of practical metrics. The US Department of Energy laid out some metrics quite a long time ago for what an electrochemical process would look like for ammonia, and they said better than 90% faradaic efficiency, [producing] around about one micro mole per square centimetre per second. So we were getting upwards of hundreds of nanomoles, so we’re getting close,’ notes MacFarlane.
Distributed solution
Based on these developments, MacFarlane has set up a spin-out company, Jupiter Ionics, that hopes to commercialise an ammonia-producing electrolyser. He envisages small devices that could run in every commercial greenhouse, producing just enough fertiliser for individual sites. These new approaches to ammonia production have the potential to empower many communities that are currently completely reliant on distant facilities and supply chains that are vulnerable to shocks from global events.
‘Suddenly fertilisers supply can be very much an on-farm concept, and that’s why our first devices can be quite small,’ he says. ‘And we have had huge interest in that concept, which only got more substantial as a result of the Ukraine war and the price hikes of gas and oil – because most fertilisers come from natural gas – and supply constraints also.’
Fertiliser supply can be very much an on-farm concept
This idea of smaller scale fertiliser producing units has gained traction in recent years. For example, as a key part of its plan to reduce greenhouse gas emissions, Japanese food company AjinoMoto has partnered with Hideo Hosono, a materials scientist at Tokyo Institute of Technology in Japan, to develop a low-temperature, low-pressure on-site ammonia production facility.
A more distributed system of fertiliser production could also bring significant economic benefits to many developing countries. In a recent analysis, Torrente-Murciano showed how Sierra Leone could benefit to the tune of $230 million (£180 million) over 30 years by using local hydropower to produce green ammonia rather than relying on imports. She believes that many other sub-Saharan nations could see similar benefits.
‘There are many countries in Africa that don’t have access to fertilisers, and that means that they put a lot of emphasis on producing food and the whole economy of these countries is based on agriculture just to feed their population. We believe that is what is limiting the real development of the country,’ she says. ‘If you have a look to what happened in Europe many, many years ago, it wasn’t until you have certainty that you’re going to be able to feed your population that people can start shifting jobs into more industrialised ones – so we think that agriculture and food is at the foundation of any developed society.’
However, producing ammonia more locally and using it widely as an energy carrier brings additional challenges in terms of safety. ‘The people who are handling ammonia at the moment are highly trained people, and if we actually want to use ammonia as an energy vector in a more distributed manner, we might need to think about very radical ways of handling ammonia – and that requires a lot of technology development,’ notes Torrente-Murciano.
Torrente-Murciano believes that much more needs to be done to increase public awareness of both the benefits and hazards associated with ammonia. ‘I don’t think that the general public is aware that ammonia is less flammable than hydrogen by far [or] that you can detect ammonia with your nose at very low levels,’ she says. ‘That is by itself very useful, because you know when there is a leak, which might not happen with hydrogen.’
‘We also need to provide expert advice and really get engaged with the public about ammonia – the goods and the bads. I think that we need to be aware of the challenges, rather than allowing people to be scared because of lack of understanding,’ she adds. ‘That is something that the whole community needs to unify forces to address.’
Jamie Durrani is a senior science correspondent at Chemistry World

















No comments yet