Designer receptors help understand cellular signals and could treat epilepsy and Parkinson’s disease, but need new tools, finds Andy Extance
What would it be like to be able to sense something that no-one else can? It might sound like a superhero comic, or a worrying delusion – but in a modest form, biochemical techniques have already made it happen. In 2005, US scientists enabled mice to taste the normally flavourless molecule spiradoline by hijacking the protein machinery that helps them sense, genetically modifying them to produce ‘designer’ receptors.1
Protein receptors are locks that, on encountering smaller molecules that serve as keys, are continually triggering biological processes in living creatures’ cells. Chemists commonly make new keys in the form of drugs – but since the late 1980s researchers have increasingly been targeting the lock too.
The scientists investigating taste processes used a modified kappa-opioid receptor designed by Bruce Conklin’s team at the University of California, San Francisco, in the US and the Gladstone Institute of Cardiovascular Disease, a non-profit research organisation in the same city.2 Replacing natural taste receptors with designer ones let mice taste spiradoline as bitter or sweet, depending on which taste cells the receptors were located in. If the receptors were in sweet cells, mice were attracted to spiradoline. If the receptors were in bitter cells, mice were repelled. Conklin calls this ‘the most elegant experiment’ using his team’s technology. ‘It’s not the biochemical signal, but the placement of the signal that causes the behavioural response,’ he says.
The technology Conklin’s team pioneered, known as receptors activated solely by a synthetic ligand (Rassls), was just the first generation. Rassls let researchers explore body functions, but met problems trying to open a window onto the brain. To do this, DNA pioneer Francis Crick wanted ‘to be able to turn the firing of one or more types of neuron on and off in the alert animal’. Scientists do that today with optogenetics, where they introduce light-sensitive proteins that instantaneously trigger neurons of interest. But improved Rassls, known as designer receptors exclusively activated by designer drugs (Dreadds), are also widely used for chemogenetic stimulation over longer periods.
It’s not the biochemical signal, but the placement of the signal that causes the behavioural response
From being used mainly in mice, chemogenetic systems like Dreadds are now progressing into non-human primates, on the path to one day being used in us. But this progress brings new challenges, exposing the limitations of existing technologies. An important ligand key works differently than thought, making current Dreadd locks harder to use outside mice. Consequently researchers must broaden their menu of synthetic receptor–drug systems. In doing so, they stand to further improve what we know about how our bodies and brains work, and potentially treat ailments like epilepsy and Parkinson’s disease.
Promise and problems
The Rassl idea originated in the late 1980s, before drug discovery scientists could get x-ray crystal structures of receptor proteins that cross cell membranes. The only way they could map how ligands bind to receptors was to clone receptors and mutate individual amino acids, explains Catherine Strader from US biotech consultants Synergy Partners. Then working at US pharma giant Merck & Co’s site in New Jersey, Strader and colleagues also modified the ligand targeting the receptor. If a binding interaction involves one group on a ligand and one amino acid, replacing one or both ‘should have the same impact’ on binding energies, Strader explains.
Strader and colleagues were studying the β-adrenergic receptor, and found that swapping an aspartic acid for serine greatly reduced how tightly natural ligands like adrenaline bound. ‘Then we had the idea of seeing if we could find a ligand that would interact with that receptor,’ she says. Eventually Merck chemists synthesised a compound that activated mutant receptors, but not natural ones.3 ‘We wrote that it would be possible to use this receptor in a transgenic animal, but never did any experiments along that line,’ Strader says.
In the following years, Conklin was inspired by Strader’s experiments and set out to make designer receptors for use as switches for biochemical processes in cell-based therapies. However, the compound the Merck team designed broke down too quickly in live animals. Conklin therefore took the idea to nearby Genentech, whose second commercial product was recombinant growth hormone made by inserting the gene for the human hormone into host bacteria. He asked whether a designer growth hormone would be a better trigger for the switch. ‘They just laughed and informed me how much money it would cost to approve a designer growth hormone,’ he recalls. But the Genentech scientists did have some good advice: stick to G-protein coupled receptors (GPCRs) like those that bind opioids, since they are activated by drugs that are readily available.
Conklin’s team first developed opioid-based Rassls and demonstrated they could be used to control heart rate.4 These Rassls would be the ones later used in the mouse taste experiments, while scientists also developed Rassls for a wide range of other receptors, using existing drugs. Conklin began hosting international Rassl user meetings, but quickly found that these designer GPCRs had shortcomings. The switches could get jammed on, bringing intrinsic ‘basal’ activity. Introducing high levels of Rassls into different tissues caused heart failure,5 massive bone deformities,6 and even a buildup of fluid on the brain known as hydrocephalus.7 Rassls’ activating drugs could also be complicated to use experimentally, since they activated natural opioid receptors too. Improvements were clearly needed.
Evolutionary impact
During that time, Bryan Roth from the University of North Carolina at Chapel Hill, US, was working on similar technology, and had telephoned Strader to discuss it. ‘I came up with the idea of doing directed molecular evolution in yeast,’ Roth recalls. This move was notable, given that Strader and Conklin’s teams tried to rationally decide which amino acids to mutate. By contrast, Roth’s team randomly generated many mutant receptors using error-prone polymerase chain reaction gene copying.
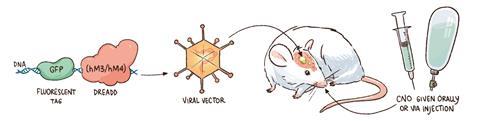
Eventually, postdoctoral researcher Blaine Armbruster developed a system using a derivative of schizophrenia drug clozapine, clozapine N-oxide (CNO), which is thought to be inactive as a drug.8 Starting from the M3 muscarinic receptor, known to bind clozapine, Armbruster selected versions activated by CNO over three generations of mutants. While the result was the first Dreadd, it bound both CNO and clozapine – a point relevant today, especially given the technology’s importance in neuroscience. ‘Clozapine is at least 10 times more potent at activating the receptor than CNO,’ Roth openly admits.
The CNO Dreadd approach has become commonplace partly because it is ‘dirt cheap’, Roth says. The virus needed to get mice to express Dreadds costs $300 for a vial that contains around 50–60 doses, he adds, and CNO costs ‘a few dollars per mouse’. Injecting virus into specific brain cells enables scientists to study the cells’ role in behaviour, for example. Such injections require stereotactic apparatus, but Roth says this is relatively common. Three or four weeks after injection ‘you give them CNO and get the behaviour’, he explains. ‘It’s very robust.’
By contrast, complementary optogenetics approaches need laser equipment, which makes the barrier to entry higher ‘in terms of cost and technical expertise’, Roth says. Additionally, while optical stimulation turns on and off very quickly, chemical stimulation lasts for longer, allowing scientists to do a different range of experiments.
At least 800 reports have already referred to Dreadds, often with profound revelations about how neurons shape behaviour. For example, researchers have used CNO Dreadds to inhibit activity at individual highly-connected ‘hub’ neurons to show their importance in memory formation.9 Another of many such experiments recently showed how neurons in the hypothalamus with high levels of receptors for the hormone somatostatin can control appetite.10
Some researchers are now looking to bring Dreadd-based treatments to people. For example, in intractable epilepsy cases, patients often have the brain area where seizures are focused removed. They could instead be given a gene therapy encoding an inhibitory, silencing Dreadd, turning off signals driving seizures. Roth notes that the concept has been proven in rodents already, and that the approach could exploit clozapine rather than CNO. ‘You could use homeopathic doses of clozapine and the advantage is that you don’t have to get approval for the drug as well,’ he explains.
Yet, ironically, when Roth has spoken with drug companies they were more focused on using CNO itself, and were worried about human bodies breaking it back down to clozapine. Mice and rats were thought not to have the enzyme that does this transformation. Other species, like guinea pigs and primates, do. ‘So they were very concerned about moving it to humans in the absence of another ligand,’ Roth says. Such concerns are being borne out as Dreadds move beyond mice to develop therapies and better understand thought processes.
Limited availability
For example, Jessica Raper from Emory University in Atlanta, US, put inhibitory Dreadds into the amygdala of two infant monkeys. Social and emotional behaviour is affected by damage to this brain region. The amygdala can also be affected in many neurodevelopmental disorders, and Raper wants to study the resulting social and emotional impacts. Inhibitory Dreadds should controllably mimic the effects of amygdala damage, without it being permanent, she explains. Giving the monkeys with Dreadds low-dose clozapine or CNO reduced emotional reactivity to human intruders.11 ‘We could also alter attention to emotional videos of another monkey or a snake,’ Raper adds.
Yet the process wasn’t as straightforward as Raper hoped. Her Emory colleague Adriana Galván was also having difficulty using Dreadds to help develop treatments for Parkinson’s disease. ‘At conferences, everyone trying to do non-human primate Dreadds had the same story,’ Raper recalls: ‘”Well, it kind of worked in two monkeys, but not in a third.” ’
Clozapine showed clear and exquisitely high affinity, potency and Dreadd engagement
Galván, Raper and colleagues therefore looked at how quickly CNO was getting into the monkeys’ brains. The amount of CNO in blood was ‘just whopping’, Raper recalls, but by comparison the amount in cerebro-spinal fluid reaching the brain was ‘so very little’. They instead found that CNO was being kept out of the brain via reverse transport, and converting into clozapine at levels sufficient to activate Dreadds.12
Mike Michaelides of the National Institute on Drug Abuse in Baltimore, US, is also seeking to develop and/or optimise ‘neuromodulation-based technologies’ like Dreadds for use in people. When he started his lab in 2015 there was no way to directly and remotely see the switches. Michaelides concluded that positron emission tomography (PET) was the best tool for the job, and started working on that approach. ‘The key first step was to characterise the precise, in-vivo pharmacology of the prototypical Dreadd ligand, CNO,’ he explans. His team therefore produced CNO labelled with radioactive atoms and studied how it interacts with Dreadd receptors, in isolated cells, rats and mice, using PET.12
‘These experiments highlighted, to our initial surprise, that CNO exhibited weak affinity, very low brain permeability, and lack of in vivo Dreadd engagement,’ says Michaelides – even in rodents. By contrast, clozapine showed ‘clear and exquisitely high affinity, potency and Dreadd engagement’. ‘Given the prior reports of CNO conversion to clozapine, and the relatively high doses of CNO that most of the research community was using, our hypothesis was that back-converted clozapine from CNO was the likely in vivo Dreadd actuator. It turned out that our hypothesis was correct as we directly showed that clozapine, converted from CNO, is the in vivo actuator of Dreadds expressed in the brain.’
Designer, but not exclusive, receptors
Michaelides therefore casts doubt on the core idea of chemogenetic switches exclusively activated by drugs that do nothing else. However, that need not undermine the important findings they’ve already provided, as long as experiments include subjects without Dreadds given CNO as controls. It has however delayed Dreadd work in non-human primates, required prior to human application, because they needed high CNO doses for the system to work. ‘[This] complicated delivery of the drug and led to side effects from high levels of converted clozapine,’ Michaelides says.
Despite the fact that we have these other non-CNO ligands, nobody uses them!
Roth is frustrated by the situation, but perhaps not for the reasons you’d expect. ‘We were aware of the potential metabolic liabilities of CNO and started creating non-CNO analogues more or less immediately,’ he says.13 ‘We published them first in 2015, but I’ve been telling people about those for probably three or four years prior to that.’ Like Conklin’s first Rassl, some new Dreadds are based on the kappa-opioid receptor, but in this case activated by salvinorin B. Another is more similar to existing Dreadds, paired with an entirely new drug known as compound 21.14 ‘Now, despite the fact that we have these other non-CNO ligands, nobody uses them!’ Roth laughs exasperatedly. He recalls applying for funding for the work, only to be told ‘CNO works great, why the hell do you need something else?’
The desire to push chemogenetics further is now driving more fundamental work on Dreadd systems, however. For example, Michaelides’ group is now developing better drugs to use with Dreadds. They have also made a PET imaging agent including radioactive fluorine isotopes. ‘Two of the many ligands we synthesised exhibited very high affinity, in vivo potency and of course in vivo Dreadd engagement,’ Michaelides says.15 These ligands worked in both rodents and non-human primates, a promising step towards use in human medicine.
While the idea of using Dreadd-based treatments may seem far-fetched, scientists like Michaelides can build on recently approved gene therapies to bring them to humans. To realise them, there will no doubt be further challenges in optimising receptor–drug pairs that fit each other and do little else. Yet taking the chemical principles routinely used in drug discovery further in this direction could one day change what we think when we hear the word Dreadd. The person talking would not be scared or apprehensive – instead the word may be linked to feeling better.
Andy Extance is a science writer based in Exeter, UK
References
1 K L Mueller et al, Nature, 2005, 434, 225 (DOI: 10.1038/nature03352)
2 P Coward et al, Proc. Nat. Acad. Sci. 1998, 95, 352 (DOI: 10.1073/pnas.95.1.352)
3 C D Strader et al, J. Biol. Chem., 1991, 266, 5
4 C H Redfern et al, Nat. Biotechnol., 1999, 17, 165 (DOI: 10.1038/6165)
5 C H Redfern et al, Proc. Natl Acad. Sci. USA, 2000, 97, 9, 4826 (DOI: 10.1073/pnas.97.9.4826)
6 J Peng et al, Endocrinology, 2008, 149, 1329 (DOI: 10.1210/en.2007-0235)
7 E J Sweger et al, J. Neurosci, 2007, 27, 2309 (DOI: 10.1523/jneurosci.4565-06.2007)
8 B N Armbruster et al, Proc. Natl Acad. Sci. USA, 2007, 104, 5163 (DOI: 10.1073/pnas.0700293104)
9 G Vetere et al, Neuron, 2017, 94, 2, 363 (DOI: 10.1016/j.neuron.2017.03.037)
10 S X Luo et al, Science, 2018, 361, 76 (DOI: 10.1126/science.aar4983)
11 J Raper et al, bioRxiv, 2018, 534214 (DOI: 10.1101/534214)
12 J Raper et al, ACS Chem. Neurosci., 2017, 8, 1570 (DOI: 10.1021/acschemneuro.7b00079)
13 J L Gomez et al, Science, 2017, 357, 503 (DOI: 10.1126/science.aan2475)
14 E Vardy et al, Neuron, 2015, 86, 936 (DOI: 10.1016/j.neuron.2015.03.065)
15 K J Thompson et al, ACS Pharmacol. Transl. Sci., 2018, 1, 61 (DOI: 10.1021/acsptsci.8b00012)
16 J Bonaventura et al, bioRxiv, 2018, 487637 (DOI: 10.1101/487637)













1 Reader's comment