Keeping astronauts alive requires some clever chemistry, as Katharine Sanderson discovers

The year is 2083 and astronauts huddle together in their launch capsule, craning to get a view of the fast-disappearing Earth. Their every breath uses up precious oxygen and produces suffocating carbon dioxide; each trip to the toilet adds a burden of waste to their cramped quarters and sees more precious water disappear. Every bite of food depletes the supplies they have on board. As they hurtle further and further from the safety of planet Earth, towards the thin, toxic atmosphere of Mars, the astronauts face the startling realisation that they are never again going to eat an apple, drink fresh water or take a deep breath of non-recycled air.
Yet those astronauts must be able to survive their three-year journey and a new life on another planet. And survive they will. They will eat, breathe, sweat, wash, defecate. They might even have some fresh food.
The technologies that will be used in the future of space flight are already being developed here on earth-bound laboratories. And it is no surprise that much of the technology used to keep astronauts alive relies on chemistry: filters, water-purifying membranes, microreactors and gas-capturing technologies.
These technologies are also being tested for future long-haul space flights in our most distant lab available: on board the International Space Station (ISS), 400km above the Earth’s surface. But the ISS has a distinct advantage over future Moon-bases or Mars missions, because even though the ISS recycles much of its water, air and rubbish there is always the option to resupply from Earth.
Nor any drop to drink
While some water is carried up to the ISS (at great expense), most of the astronauts’ supply comes from recycling their sweat, urine and washing-up water. To do this, the chemists’ old friends distillation and filtration are used. But even these things become more complicated in space: astronauts’ sweat and urine isn’t the same as everyone else’s. In space, we shed a little more calcium from our bones than we do when our bones are under the stress of gravity. The water-purification systems developed here on Earth include sulfuric acid – there to kill off micro-organisms – which in space combines with the extra calcium to make calcium sulfate. This precipitates out and clogs up the distillation and filtration equipment, making the system less efficient than when it is tested on the ground. ‘You can’t always anticipate everything,’ says Darrell Jan, an air revitalization researcher at Nasa’s Ames Research Center in California, US. Nasa researchers are now working to tweak the purification additives to see if the extra calcium can be worked around.

Of course, water isn’t the only essential for life. At Ames, researchers are also looking at the air astronauts breathe and the solid waste that surrounds them. Much of the astronauts’ life is like ours, with lots of plastic and food waste. That waste not only takes up precious room in a crowded space craft, it also contains items that are likely to become dangerous if left in a bin bag. ‘If you stored kitchen trash without taking it out for three years it’s going to go mouldy, it’s going to stink. It will be a hazard,’ says Jan. But in space, the food waste, plastic packaging and general trash that goes into a dustbin is a vital resource. Jan is part of a team aiming to recycle this waste usefully. The Ames team is developing a heat melt compactor, where waste goes in and plastic tiles that can be used as radiation shields on the space craft come out.

The compactor will strip the useful water out of the waste – which includes food, wet wipes and duct tape – by heating the trash and boiling the water away for collection, a process that also kills any lurking microorganisms. Further heating sterilises the mixture, then the remaining plastics are melted and compacted. During the heating, different volatile compounds are released, including aldehydes, ketones, furans, alcohols, aliphatic hydrocarbons, aromatic hydrocarbons and sulfides. Any hydrocarbons in this mixture are oxidised using platinum catalysts, and then passed over an adsorbent bed, to keep the levels of these volatiles in the cabin to a minimum.
The plastic tiles that come out of the compacter have lots of hydrogen in them, which is a bonus – they are destined to be used as radiation shields and hydrogen doesn’t produce any secondary radiation. Currently being tweaked to work in microgravity, the system shows promise as a useful way to deal with rubbish in space, and even if it looks bulky, that won’t matter on a mission to Mars. ‘The heat melt compactor is 100kg. If we saved 200kg of water, that’s a net plus,’ says Jan.
Clearing the air
The air in the cabin is also a major focus for recycling. ‘Imagine duct-taping yourself into a room,’ says John Hogan, another Nasa air-revitalisation researcher at Ames. ‘Carbon dioxide builds up very quickly and oxygen is depleted.’ All because of humans’ need to breathe; it’s the only way to get oxygen into our bloodstream and our heart pumping. But respiration is a chemical reaction: the oxygen we breathe in is used to convert glucose in our body into energy, in the form of ATP, breathing back out carbon dioxide and water. The water will be useful, but what of carbon dioxide? ‘We would like to get oxygen out of carbon dioxide,’ says Hogan.
We’re always looking where the molecules are going
On the ISS at the moment, astronauts can breathe easy thanks to a molecular sieve with four compartments, or beds, which trap the gases. Two of these are desiccants, and the other two are zeolites which absorb water and carbon dioxide. The carbon dioxide is then passed through a catalytic system, following the Sabatier reaction, where it reacts with hydrogen over a catalyst to make methane and oxygen. The oxygen is immediately useful, but the unwanted methane is currently vented out to space.
But for a Moon base, or a Martian journey, this is incredibly wasteful. ‘We’re always looking where the molecules are going,’ says Hogan. ‘We really don’t want to lose any molecules.’ Each person produces a kilogram of carbon dioxide per day, Hogan says, so a crew of four produces 4kg of carbon dioxide every single day. ‘That’s a significant amount of material and a significant amount of carbon,’ he says. Scientists are furiously working out how to make use of this onboard resource and, in future, how to make use of a Martian atmosphere, which is 96% carbon dioxide.
That suffocating carbon dioxide first needs to be captured, borrowing technologies developed on Earth for carbon capture and storage: metal-organic frameworks (MOFs) and porous coordination frameworks. In carbon capture applications here on Earth, MOFs have been looked at for their amazing ability to hold huge amounts of the gas in their mind-bogglingly large internal surface area, locking it away forever. But researchers from Nasa’s Ames research centre and the University of Puerto Rico have been looking at different MOFs – that not only take up the carbon dioxide, but can also release it again. This adds a new dimension to the development of these porous materials. ‘Many adsorbent materials display exceptional CO2 storage capacities, but not many are capable of delivering the gas in the quantities needed and without the need of energy input,’ says Arturo Hernandez-Maldonado from the University of Puerto Rico, who is part of the MOF team.
‘MOFs have set new benchmarks for the design of effective adsorbents. I think that there is a very good chance that we might end up having these materials in space cabins in the near future,’ says Hernandez-Maldonado. The team has shown that copper-based MOFs with pyrazinedicarboxylate, bipyridine and 1,2-di-(4-pyridyl)-ethylene groups are able to take up and release carbon dioxide at moderate pressures – another important factor in any space-based system.
We can re-envision the life cycle of carbon
Once that carbon dioxide has been gathered, it can be processed, again using the Sabatier reaction. The resulting methane can then be further treated to make more hydrogen or pyrolised to give larger carbon-based monomers, or even polymers. There then lies a tantalising possibility that ‘waste’ carbon dioxide could be turned into a bioplastic, to be used in a space-bound 3D printer to provide the astronauts with a resource to make the stuff they might need unexpectedly for repairs or useful gadgets. The chemist’s imagination could run wild here – even allowing drugs to be made in space – if the methane can be processed to provide the chemical building blocks for organic synthesis. ‘We can re-envision the life cycle of carbon,’ says Jeff Lee, another Ames team-member.
Biological bias
Another way to use methane and carbon dioxide is as nutrients for algae that could then be used to feed crew members. And the power of algae is being harnessed in an ambitious European project, which is looking at the astronaut life support system as a whole. The Melissa (micro-ecological life support system) project is all about making a fully life-supporting modular system, adaptable to any space-based setting, be that a spaceship, Mars or the Moon. The project is run by the European Space Agency and has been going for 25 years already, according to Melissa’s project leader Christophe Lasseur, based at the European Space Research and Technology Centre in Noordwijk, the Netherlands. ‘The idea was that Europe should be nicely positioned for future space missions after the ISS,’ Lasseur says.
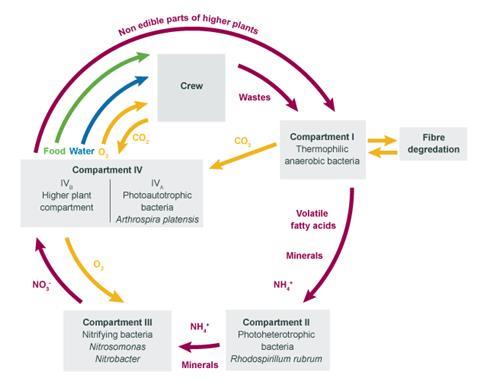
Humans in space will require around 5kg of metabolic needs – food and water – per day per person, according to Lasseur. ‘This really means to just stay alive,’ he adds: no washing, no luxury. Adding these figures up for a three year journey to Mars gives you an answer of tonnes, says Lasseur. Melissa has much in common with a lot of the ongoing research for future space outposts, in that it turns to biological systems for inspiration. Much of Melissa’s technology is focused on bioreactors, using photosynthetic microorganisms to convert carbon dioxide to oxygen.

The Melissa project is using a chemical engineering approach, something that Claude-Gilles Dussap at Université Blaise Pascal in Clermont-Ferrand, France, has pioneered. ‘Mars and the Moon don’t have the same constraints as on a flight in terms of physics and mass, so you must develop a generic approach,’ he says. And the closed-loop system that has to have all inputs and outputs balanced carefully is music to the ears of a chemical engineer like Dussap who has also embraced biology. For any life support system, chemistry can only get so far, he says. ‘You cannot produce food by a solely chemical approach, you have to use biology,’ he says.
The Melissa system will have five compartments, and a pilot plant is being developed at the Universitat Autònoma de Barcelona in Spain. The first compartment takes food and human waste and uses thermophilic anaerobic bacteria to munch it up, producing liquefied volatile fatty acids and gaseous carbon dioxide (which will be used later in the loop) plus ammonium. The second compartment is populated by light-harvesting bacteria Rhodospirillum rubrum which get rid of those fatty acids and leave just minerals and ammonium. These are fed into compartment three, which has nitrifying bacteria to turn that ammonium into nitrates, to use as feed for plants.
Rocket salad
And that’s where Melissa’s fourth compartment comes in – food. One half of the compartment is to grow the protein-rich cyanobacteria spirulina, which feed on carbon dioxide, minerals and nitrates that come from the previous compartments. But while protein is an important part of the human diet, the good news for astronauts of the future is that the other half of compartment four is essentially a greenhouse growing plants to eat. Not for these astronauts a diet of just spirulina, as Melissa will provide lettuce, wheat, rice, potatoes, tomatoes and other foods. This compartment is also where oxygen is produced, from photosynthetic bacteria.
Finally, to complete the loop, the crew will occupy the fifth and final compartment. This section isn’t yet ready for human habitation, but an approximation where rats are living happily in a smaller version is up and running with an air-recycling system in place. Lasseur says that the rats are perfectly comfortable breathing their own recycled breath. ‘The rats are very well treated, everything is done according to European ethical rules,’ he says. ‘They only complain when there is no internet connection,’ he jokes. The rats’ habitat is airtight and connected to a photobioreactor, says Lasseur, where photosynthetic bacteria convert carbon dioxide to oxygen, which is fed back to the animals. A system of filters and columns removes contaminants.

It’s this modular system of linked compartments that allows Melissa to be – theoretically – applied to a range of scenarios, according to Dussap. Previous approaches to life support systems have been ‘more like an art’, he says, with the hope that if the right biological systems were put together into a closed system, nature would evolve into a working system, he says. ‘We had to say “No, it is a chemical engineering problem”.’
One of the key problems for the Melissa and Nasa teams to solve is how to separate gas and liquids in microgravity, where bubbles don’t sink or rise in the same way they do on Earth. For this, membrane filtration technology is going to be a key component, says Dussap. The material will need to be as hydrophobic as possible, to prevent water condensing, and should also be very gas-permeable. This is just one example of technologies that will need to be developed to get a fully-functioning Melissa set-up off the ground (literally).
Just when that might happen depends on how much manpower can be thrown at the problem, says Dussap. He hopes that by 2020, on Earth at least, a prototype that could support humans will be ready. And then as soon as 2030, thoughts could perhaps turn to a Moon or Mars base, he says.
And as those future-world astronauts settle into their cramped quarters, their thoughts are likely to be focused on the task ahead of them. As they take their last breath of fresh Earthly air, will they be contemplating the MOFs in their air filtration system? The fibre optics being tweaked to provide the right amount of light for the bacteria breathing in their carbon dioxide? Will they be mulling over the chemistry that will let them drink and breathe their own recycled effluent for years to come? It seems more likely that as they lie back, watching their home disappearing, the last thing on the mind of those travellers with a one-way ticket is going to be the chemistry keeping them alive.
Katharine Sanderson is a science writer based in Derbyshire, UK
UPDATE The graphic showing the Melissa lifecycle was corrected on 12 September 2016












No comments yet