Food companies are starting to switch from synthetic to natural sugar substitutes, Rachel Brazil reports

Our addiction to sugar is fuelling an epidemic of obesity and diabetes in the developed world. This has led to the use of synthetic zero-calorie sweeteners to reduce our sugar consumption, but in the last decade, rightly or wrongly, they have received a lot of bad press. Instead, food and drink companies have started to look at natural alternatives for the $7.4 billion (£5.0 billion) sweetener market and the first plant-based products are now making their way onto supermarket shelves. In 2014, the Coca-Cola Company launched Coca-Cola Life, made with a blend of sugar and stevia leaf extract and containing 37% less sugar than regular Coke. With its green packaging, in contrast to Coke’s traditional red, the message is that with natural non-caloric sweeteners, even the health-conscious consumer can indulge their sweet tooth.
Although limited in number, sweet compounds are chemically diverse, with at least 50 structural classes represented. The first synthetic sweetener was saccharin, discovered in 1878 and the first product of Monsanto (now a multinational agrichemical company). It is 300 times sweeter or, more correctly, 300 times more potent that sucrose (because it does not taste sweeter, but can start to be tasted at lower concentrations). It was initially developed to compensate for sugar shortages during the first world war, but took off in the 1960s when its taste was found to be improved by blending it with another artificial sweetener, cyclamate, discovered in 1937. From the 1960s onwards, chemists discovered other high-potency sweeteners, such as aspartame, sucralose and most recently neotame in 1992. Today, aspartame is the most widely used high potency sweetener and particularly when blended with other sweeteners, comes closest to reproducing the taste of sucrose.

Questions have been raised over the safety of several artificial sweeteners. In 1969, after experiments linked the saccharin–cyclamate blend to bladder cancer in rats, the US Food and Drug Administration banned cyclamate and in 1972 removed saccharin’s ‘generally recognised as safe’ status – not declaring it safe again until 2001. Both cyclamate and saccharin are approved by the European Food Safety Agency (EFSA). And although aspartame has been approved in both the US and EU for almost 20 years, it is still branded unsafe by several consumer advocacy groups. Disputed experiments linking it to brain tumours in rats and internet conspiracy theories on the validity of the original safety data have been compounded by a spate of hoax emails claiming the sweetener causes numerous diseases. A 2013 EFSA review concluded it posed no toxicity concerns.
Grant DuBois, a sweetness technology consultant and former director of ingredient and product sciences at Coca-Cola, says aspartame is incredibly safe. ‘It says something about human psychology – if something appears too good there must be something wrong. There just never has been anything wrong with it.’ On consumption, aspartame is rapidly metabolised into phenylalanine, aspartic acid and methanol – the first two are found in vegetable and animal protein and the last in fruit and vegetable complex carbohydrates.
Going natural
But regardless, the food industry cannot ignore overwhelming public perception, whatever its basis. DuBois says that as early as 2000, Coca-Cola started to look at natural sweeteners. Finding new commercially viable sweeteners, synthetic or natural, is not easy and during his 20 years at the company, DuBois says he was constantly approached by companies with new sweeteners which were invariably unsuitable. He created a list of metrics which includes safety, stability, solubility, cost effectiveness and above all taste.1 For something to be commercially viable, DuBois says ‘you really have to hit pretty high on most of these metrics’. In general, the known natural sweeteners do not score as highly as currently used synthetic sweeteners.
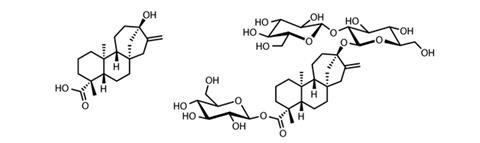
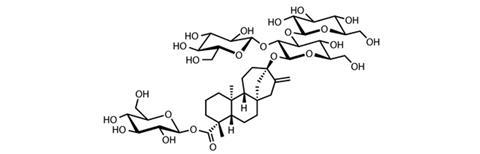
One of the best natural non-caloric sweeteners and the first on the market (approved in 2011 in the EU and with a ‘generally recognised as safe’ status in the US), comes from the leaves of the Stevia rebaudiana shrub. Found in Brazil and Paraguay, the stevia plant has been used by the local Guarani people for centuries. Its sweetness comes from a series of steviol glycosides – molecules based on the diterpene steviol with differing additional glucose and other sugar groups attached. The structure of the main sweet molecule, stevioside, was elucidated in 1955, but the latest studies have found the plant to contain more the 40 distinct steviol glycosides. This includes rebaudioside A – whose taste is most similar to sugar, but is 200 to 300 times more potent as well as being heat and pH stable.
Rebaudioside A is the main component of commercially available products, but these are standardly blended with non-caloric ‘bulk’ sweeteners – sugar alcohols such as erythritol that are half as sweet as sugar but provide a blend that better mimics the taste, texture and ‘mouthfeel’ of sucrose. Erythritol itself is naturally derived from fermenting corn starch (although a recent successful US law suit prevents it being marketed as ‘natural’).
Products containing steviol glycosides are entering the market quickly. Coca-Cola’s lemonade brand Sprite now contains steviol glycosides and 30% fewer calories, Tate & Lyle has produced a sugar–stevia sweetener blend it calls Light at Heart for baking, and stevia sweetener is being used in some yoghurt and chocolate. Blending steviol glycosides with sugar allows significant calorie reductions without too great a loss in taste quality.

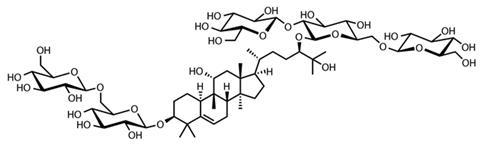
The other natural non-calorie sweetener making its way onto supermarket shelves comes from the monk fruit or luo han guo – the fruit of the Siraitia grosvenorii vine, native to southern China and northern Thailand. The fruit is 300 times more potent than sucrose and was used in Chinese medicine to treat sore throats. In the 1980s, Japanese food scientists began commercial development and the sweet compounds were identified as a group of glycosides called mogrosides all with a triterpenoid structure. The sweetest of these is known as mogroside V or esgoside. DuBois says that Coca-Cola did work on the monk fruit sweeteners, but the cost of extraction and purification was too high. Several companies have produced a monk fruit extract, including Tate & Lyle who launched its Purefruit brand in the US and Asia in 2011.
Sweet and bitter
You might expect there to be several types of sweet taste receptors given the range of molecular structures that taste sweet, plus the fact that we have 25 different types of bitter taste receptors. But according to food chemist Gabriella Morini from the University of Gastronomic Sciences in Pollenzo, Italy, this is not the case. The structure of the sweet taste receptor in our taste cells was identified in 2001.2 It is made up of two G-protein-coupled receptor (GPCR) proteins – large proteins embedded in the cell membrane, winding across it seven times, so that the protein sits permanently partly inside and partly outside the cell. Binding sweet molecules outside the cell triggers secondary messaging inside the cell, which sends signals to the brain. Sucrose and other sugars are bound to a large region of the receptor protein outside the cell, described as a Venus fly-trap domain due to its shape and change in conformation on binding a carbohydrate. But Morini says there are other secondary binding sites that can activate the receptor. These allow larger molecules to trigger a response without the need to fit into the deep cleft of the Venus fly-trap and this explains why so many different types of molecules can taste sweet.
One of the problems with high-potency sweeteners, including steviol glycosides, is their bitter aftertaste. Binding to bitter taste receptors accounts for some of this effect, but according to Morini ‘why most of the intensive sweeteners have a liquorice aftertaste has not been understood yet’. Her work has also shown a significant genetic variation in the sensitivity to the bitter aftertaste of stevia. Morini and colleagues tested the perception of its taste in 100 volunteers; to some it tasted sweet and bitter, to others only sweet. They went on to identify the two single nucleotide variations (known as single nucleotide polymorphisms or SNPs) in the genetic code of the two bitter taste receptors that are responsible for the differing stevia taste perceptions.3
Turning sour to sweet

Perhaps the strangest twist on non-caloric sweeteners is the miracle fruit, the berry of the west African plant Synsepalum dulcificum. It contains a protein named miraculin due to its miraculous properties. It is itself not sweet, but causes subsequently eaten sour acidic foods such as citrus to taste sweet. The fruit was a popular novelty food in the 1970s and recently it has been taken up again by high-end chefs such as ‘molecular gastronomer’ Homaro Cantu of the Moto restaurant in Chicago, US. He uses it for what he calls ‘flavour-tripping’ and is looking at how to integrate the berry powder into foods such as doughnuts, which can then be made without sugar.
The 191-amino-acid protein was isolated in 1968 but an explanation of how it works was only published in 2011 by a team led by Keiko Abe from the University of Tokyo, Japan.5 Miraculin is thought to bind strongly to the sweet taste receptor without activating it until the pH drops. When acid is introduced, the protein changes conformation and is able to turn the receptor on, creating a strong sweet taste which can drown out the sour. The effect generally lasts for about an hour, while the miraculin protein is still bound to the receptor.
Attempts to commercially develop miraculin continue in Japan where, unlike the US and Europe, it is approved for use as a food additive. Rather than just a novelty, it has been marketed as a diet product and alternative to sweet foods. Biotechnologist Hiroshi Ezura from the University of Tsukuba is developing a transgenic route for cheaper production, and has expressed the protein in Escherichia coli, tomatoes and lettuce. Japanese horticulturalist Mitsuharu Shimamura has succeeded in cultivating the plant for mass production, which needs temperatures of above 20°C to produce fruit. Once harvested, the miracle fruit can be consumed as they are, or dried, powderised and turned into tablets (which are available for purchase online). Cafes have now opened in Tokyo and Osaka selling food to go with the fruit – customers are instructed to chew on the berry before tucking into the sour cakes and desserts on offer.
Sweet proteins
An unusual category of natural low-calorie sweeteners are proteins found in a number of tropical fruits, some more than 1000 times more potent than sugar. A number of these proteins were investigated in the 1970s, such as thaumatin, found in the west Africa katemfe fruit, which was developed by Tate & Lyle, but large-scale use never took-off. Neurophysiologist, Goran Hellekant, from the University of Minnesota Duluth, US, was involved in some of the early work on another sweet protein, brazzein. His work started in the 1990s with an encounter with French ethnobiologist, Claude-Marcel Hladik, who was studying the eating habits of apes in west Africa and came across the berry of the Pentadiplandra brazzeana plant. Locally it was called j’oublie (meaning I forget), because the red fruit was used in weaning infants to help them forget the taste of milk by attracting them to the sweet-tasting fruit. Hellekant’s group were eventually able to isolate the 54-amino-acid protein brazzein, which along with another protein pentadin is responsible for the sweetness.4 Hellekant says brazzein is stable and has no bitterness.
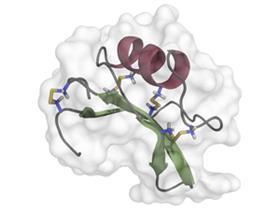
Rather than attempt to cultivate the plant, Hellekant and colleagues developed methods to express brazzein in bacteria and yeast. ‘Being the best natural sweet protein when it comes to the taste, it seemed to me that it should have had a pretty good future,’ says Hellekant, and there are ongoing attempts to market it, using the brand name Cweet. But as DuBois points out, sweeteners manufactured in genetically modified organisms also suffer from negative public perceptions and this defeats the original rationale for natural sweeteners.
The other major weakness in the taste of protein sweeteners are their temporal profile – how long it takes for the sweet taste to develop once it enters the mouth and how long it lingers. Compared to sucrose they are slow to taste sweet with a lingering aftertaste that often feels cloying and unpleasant. The effect is due to the sweetener binding to the mucous membrane lining inside the mouth which causes a delay in the maximum sweetness intensity. This also causes the lingering effect, as DuBois explains. ‘The sweetener comes off the receptor and unlike sugar which just washes down your throat, sweeteners will bind again to the cell membranes, staying around to activate the sweetener receptors over and over.’ Whilst the effect is seen in other high-potency sweeteners, it is pronounced in sweet proteins due to their hydrophobic nature.
The wider function of taste
While its taste quality may not be able to match the synthetic alternatives, stevia is projected to account for 15% of the global sweetener market by 2020. DuBois now uses his expertise to consult on improvements to stevia-based products, and is convinced that non-bitter rebaudioside A formulations without sweetness linger are soon to come. But recent findings are leading to questions over the whole concept of replacing sugar with low-calorie sweeteners, synthetic or natural. Some studies have indicated that low-calorie sweeteners do not help people lose weight and we may be starting to understand why.
It would be wise to get used to less sweet food
In 2007, sweet taste receptors were discovered in the stomach and intestines and it became clear that our taste receptors have a wider metabolic role.5 They allow us to detect sugar and regulate our capacity to absorb it through the intestines, via glucose transporter proteins. These receptors will of course respond to all sweeteners. The suggestion is that stimulation by non-caloric sweeteners could affect the body’s glucose balance and other metabolic processes including insulin release. ‘There are studies going on to understand if different sweet compounds, natural or non-natural, are able to have a different effect in non-sensory locations,’ says Morini, adding that no firm conclusions can be drawn until these are reported. Another interesting, albeit disputed study, suggested in 2014 that several artificial sweeteners altered the metabolisms of mice and linked the effect to changes caused to gut bacteria.6
Rather than stimulating our sweet taste receptors with non-caloric sweeteners, Hellekant suggests that the answer to obesity is to dampen our response to sweetness. He has created transgenic mice that are unable to transmit the nerve impulses from sweet taste receptors to the brain and has found these mice eat less and weigh 30% less than their sweet tasting counterparts. ‘Those who do not taste sweet live significantly longer – the difference is really striking,’ says Hellekant. He thinks that finding a pharmaceutical way to reduce stimulation of the sweet taste receptor could be a future therapeutic strategy for treating obesity.
Evolutionarily speaking, taste has been very important – it allows us to do a kind of instant chemical analysis to find calories quickly. But Morini points out our senses have evolved less rapidly than the way we prepare and eat food. She suggests that now ‘it would be wise to get used to less sweet food and learn to like foods that have a different taste’. Whilst it seems unlikely we will lose our taste for sweetness, we also possess taste responses designed to push us towards micro nutrients, such as the bitter, hot or astringent compounds found in vegetables and these are often enjoyed more by those accustomed to eating them.
Rachel Brazil is a science writer based in London, UK
References
1 G E DuBois, ACS Symp. Ser., 2009, 979, 444 (DOI: 10.1021/bk-2008-0979.ch029)
2 G Nelson et al, Cell, 2001, 106, 381 (DOI: 10.1016/S0092-8674(01)00451-2)
3 D Risso et al, Genes Nutr., 2014, 9, 401 (DOI: 10.1007/s12263-014-0401-y)
4 D Ming and G Hellekant, FEBS Lett., 1994, 355, 106 (DOI: 10.1016/0014-5793(94)01184-2)
5 H-J Jang et al, Proc. Natl Acad. Sci. USA, 2007, 104, 15069 (DOI: 10.1073/pnas.0706890104)
6 J Suez et al, Nature, 2014, 514, 181 (DOI: 10.1038/nature13793)
7 A Koizumia et al, Proc. Natl Acad. Sci. USA, 2011, 108, 16819 (DOI: 10.1073/pnas.1016644108)












2 readers' comments