Chemists who want to make materials that repel water but do not contain fluorocarbons are taking their inspiration from nature, Rachel Brazil finds
Chemists are seeking alternatives to organofluorine compounds for non-stick coatings. Teflon, polytetrafluoroethylene, has been coating our frying pans since the 1940s. But with increasing evidence of organofluorines’ environmental persistence, bioaccumulation and toxicity, the hunt is on for new non-stick solutions. One place where chemists have been looking enviously is nature itself. Numerous plants and animals have surfaces designed to repel water, and in some instances even oil. But the chemistry of these surfaces is not nature’s only trick – complex nanostructuring plays a huge role. Exploiting these designs is now helping chemists to produce the next generation of non-stick coatings.
The wettability of a surface is measured by the contact angle of a drop of liquid on the surface. This essentially measures how much of the drop is in contact with the surface – the higher the angle the smaller the contact with a surface. In the case of water, when this angle exceeds 150°, the surface is considered superhydrophobic and very difficult to wet.
There is an enormous range of substances that can be used
Biologist Wilhem Barthlott at the University of Bonn in Germany has made some of the major breakthroughs in understanding superhydrophobicity in biology over the last 30 years. He suggests that superhydrophobicity must have evolved 450 million years ago when life moved onto land and started respiring via gas exchange. ‘We have looked at about 24,000 different species of plants and animals and many of them are superhydrophobic or at least have superhydrophobic parts,’ he says.
Many extrude a crystalline waxy layer. Barthlott says they are usually complex mixtures of long-chain hydrocarbons (20–30 carbons) which can be alkanes, ketones, aldehydes, alcohols, fatty acids and esters.1 But it’s not only the chemistry that reduces wettability. ‘There is an enormous range of substances that can be used, but they would never reach the quality of a Teflon coating,’ explains Barthlott. ‘The secret of life was to invent a complex, highly sophisticated hierarchical structuring of the surface.’ The surfaces form crystal structures such as tubules, platelets or threads, but these cover another textured layer of cells or hairs creating two or three superimposed levels of ‘hierarchical sculpturing’ at the micro and nano scales. ‘The only thing forbidden is flat!’ says Barthlott.
Sticky or slippery
The function of the rough hierarchical surface is to create air pockets. A water droplet sits on top of the trapped air and this drastically reduces the contact between solid and liquid, allowing the droplets to form near perfect spheres which easily roll off. One of the most famous examples of this behaviour is the lotus leaf, whose self-cleaning surface has a contact angle approaching 180°. Scanning probe microscopy shows the leaves are covered with 1–5μm bumps called papillae underneath a waxy crystalline top layer. This means rain drops will roll off, along with any surface dirt.
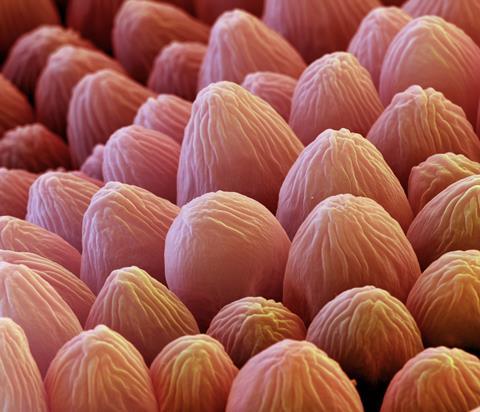
Another example from biology is the rose petal. Its surface is also hydrophobic, but it behaves very differently. ‘When you put a drop of water on a rose petal you must have noticed that the water sticks on the surface – it doesn’t fall off. It’s a sticky hydrophobicity,’ explains Ayyappanpillai Ajayaghosh, a chemist at the National Institute for Interdisciplinary Science and Technology (NIIST) in Thiruvananthapuram, India. Ajayaghosh has tried to mimic the surfaces of both the lotus leaf and the rose petal.
As with the lotus leaf, the rose petal has a hydrophobic wax layer, covering a hierarchically textured surface formed from nanofolds in its cuticle and conical shaped papillae. But the difference is the size of these structures – 16μm in diameter compared to 11μm for the lotus leaf. This means that water is able to penetrate the structured surface, creating a greater solid–liquid interface and thus the stickiness. The pinning is known as the Wenzel state, whereas the drops that roll off the lotus leaf are in the Cassie–Baxter state, named after the scientists who defined these different wetting phenomena.
So can these states be replicated chemically? Ajayaghosh started with alumina – an inherently hydrophilic material.2 His team had been working on covalent-organic frameworks, a class of crystalline porous polymers, and realised that these materials could self-assemble into hydrophobic coatings. They coated the alumina surface with an azobenzene based ligand (AzPBA) and then covered this with an aromatic bis-aldehyde (BA) with two alkoxy chains, approximating the effect of a waxy coating. The contact angle reached a near-superhydrophobic 145° and the surface showed ‘sticky’ rose petal-like behaviour.
Ajayagosh then treated the AzPBA-covered surface with zinc ions before adding the BA layer. The contact angle of the new surface increased to 165° and the surface became slippery like the lotus leaf. ‘When you [add] the zinc ions, it forms a [coordination] polymer on the surface – a kind of network where there is a [new] nano-architecture,’ says Ajayagosh. The zinc ions become attached to the AzPBA carboxylic acid groups and the new surface has finger-like 1μm protrusions, 200nm wide. This is enough to trap air below a water droplet placed on the surface. Ajayagosh says the work has shown that it is quite possible to chemically mimic the superhydrophobic states found in nature by creating differing surface morphologies. ‘We are not using any fluorinated chemistry – this is very simple chemistry,’ he adds.
The omniphobic springtail
While nature can master the superhydrophobic surface with relative ease, it is far more difficult to create superoleophobic surfaces that can also resist oils and superomniphobic surfaces that resist all liquids. Chemists usually resort to fluorinated polymers, but nature doesn’t have this option. Nevertheless there are a few examples of omniphobic surfaces in nature, such as the collembola or springtail – a small wingless soil-dwelling insect. ‘The animal is exposed to highly contaminated [water] with surface active substances, so in consequence it has a low surface tension and wets a surface much easier than [clean] water,’ explains Carsten Werner from the Max Bergmann Center of Biomaterials in Dresden, Germany. ‘There is particle friction and mechanical force acting on the skin and therefore a solution has evolved that is a much more exciting template for us to copy.’
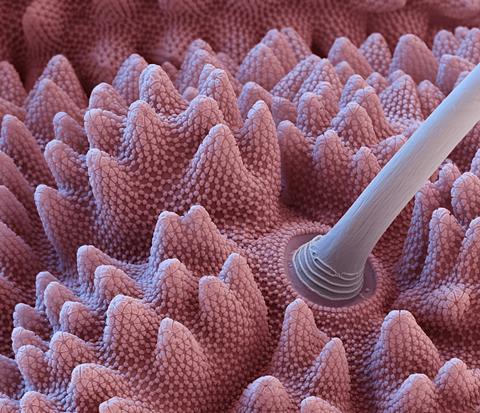
The springtail surface has highly ordered rhombic or hexagonal honeycomb-like patterns, composed of three different hierarchical layers.3 The resulting structure forms nanocavities (0.3–1μm) covering the entire body, but in cross section there are characteristic, mushroom-shaped overhangs. ‘This particular cross-sectional structure keeps nanosized air bubbles in the structure and this retention of the gas avoids wetting even of non-polar liquids,’ says Werner.
‘We were able to produce structures that really faithfully recapitulate the nano morphology of the skin, however of course limited to pretty small samples,’ says Werner. Their replicas were made by taking direct mouldings from the insect skin using polyethylene glycol diacrylate. They made two versions – one a faithful replication of the surface, the other without the nanostructured features. They found that the unusual overhanging nanostructures were key to producing a superomniphobic surface – the samples produced without it had contact angles close to zero and those with, up to 150°.4
‘The omniphobic properties we have seen on these animals depend just on the nanomorphology of the surface not on the chemistry of the materials we are using – we have demonstrated that it’s possible to achieve this effect in entirely different types of materials,’ says Werner. The overhangs seem to provoke an energy barrier that cannot be overcome even by a low-surface-tension liquid phase such as hexadecane (which was able to wet a Teflon sample).
Werner is keen to exploit these surfaces as antibacterial coatings, for example to protect surgical implements. ‘The morphology we take from the springtail by itself is quite effective and we are now working to combine these structured surfaces with coatings that contain antiseptics.’ His Dresden colleagues have been investigating laser-based lithography methods to mimic the springtail features on metals, polymers or ceramics. In 2018, a team from South Korea combined nanolithography and a wrinkling method that shrank parts of the polymer surface to fabricate a highly repellent artificial springtail surface able to endure extreme pressures. They concluded their fabricated system was superior to the insect skin itself.
Underwater

In many instances nature has used superhydrophobicity to allow plants and insect to survive under water for long periods of time. One example is Salvinia molesta, an extremely invasive fern that can survive underwater for weeks, continuing to photosynthesise. ‘It has the most complex surface we know in plants,’ says Barthlott. ‘No material scientist in their weirdest nightmare would have thought of such a solution!’ Its water-repellent surface holds a protective air layer via an array of whisk-shaped hairs (called trichomes) that make up the surface. The tips of the whisks are chemically distinct, being hydrophilic, and this firmly pins a water layer to the surface with air trapped underneath. The pinning effect keeps the air layer – as large as 3.5mm – under a negative pressure in small individual pockets.
Barthlott and colleagues at the University of Rostock in Germany have been looking at how a synthetic version could be used to coat a ship’s hull for drag reduction. Given that shipping is responsible for roughly 3% of global greenhouse gas emissions (1 billion tonnes of carbon dioxide per year) a reduction could have a significant impact . Barthlott isn’t prepared to give the details of their new material, but others have started to create similarly designed surfaces. A team of Italian engineers recently used 3D laser lithography and a hydrophilic epoxy-based photoresist to form an array of 7μm hairs that mimicked an S. molesta leaf.5
The impact of such a coating on underwater drag could be maximised by combining it with technologies such as Mitsubishi’s air lubrication system, patented in 2010 . Here compressed air forms microbubbles. ‘If you have a superhydrophobic surface it would attract [these] air bubbles like a magnet and hold them, so this could be an ideal combination,’ says Barthlott. He thinks their current prototype coating could reduce drag by up to 30%.
A chemical plastron for separating oil
Air-trapping surfaces are also common in aquatic insects. A series of tiny hairs or bumps, known as setae or microtrichia, trap a thin layer of air that allows the insect to breathe underwater – essentially acting like an external gill. This feature is known as a plastron. ‘In Texas, entire colonies of fire ants will link up and form these floating rafts with plastrons trapping [air] between them [to enhance their buoyancy],’ says materials chemist Sarbajit Banerjee from Texas A&M University in the US. He has replicated this principle to create superomniphobic surfaces.

Repelling oil is always harder than water, explains Banerjee. ‘All that holds oil molecules together are London dispersion forces, so the propensity of oil drops to spread is much higher.’ The hydrocarbon waxes found in nature are generally easily wetted by oils.
So to design a superomniphobic surface with plastron-like properties, Banerjee turned to zinc oxide nanotetrapods spray-coated onto stainless steel. These four-legged nanocrystals form when zinc foil is rapidly oxidised in air. ‘No matter how you put them down, one or two legs come up at you; you can’t form something flat,’ says Banerjee. The nanotetrapods are bonded to the steel surface using tetraethylorthosilicate, which creates a silicon dioxide link. ‘The network of tetrapods are trapped there and when we immerse [the surface] in water you see this glistening, corresponding to air bubbles that have been trapped,’ he explains.
But to replicate a superolephobic surface, Banerjee also functionalised the surface with perfluorooctane phosphonic acid (C8H6F13O3P).6 The non-polar fluorine-containing monolayer provides an even lower surface energy. ‘We essentially have C–F bonds protruding out, interacting with the water or oil molecules and so that gives us oleophobicity as well as hydrophobicity stemming from both chemistry and the texture,’ he explains. ‘The roughness acts to amplify the intrinsic chemistry.’
These materials’ superoleophobiciy makes them ideal for coating oil storage and transportation equipment, but Banerjee is particularly interested in using them for separating viscous oils from water. Large amounts of crude oil are now extracted from reservoirs using injected steam, but separating the resulting emulsion is difficult and requires materials that can function above 130°C.
Banerjee has created an entirely new filtration process based on a stainless steel mesh coated with zinc oxide nanotetrapods. ‘By itself it is quite hydrophobic, because of the layer of air [on the surface], but it’s also oleophilic,’ he explains. The mesh membrane forms an interconnected porous plastron network which lets oil through. But it keeps water droplets suspended above the air pockets formed between the protruding nanotetrapods. The water droplets are in the Cassie–Baxter state, in contrast to oil drops that are in Wenzel mode and permeate the mesh. The filter can reduce the water content of viscous oil to as little as 0.69% by volume.7
Slippery approach
Another innovative approach to omniphobic surfaces that does not rely on an air pocket came from the lab of Joanna Aizenberg at Harvard University in Massachusetts, US, and also takes it inspiration from nature. The insect-eating pitcher plant captures its prey using a slippery water-lubricated surface that repels oils found on insect feet. The lubricant itself is held in place by an irregular micro-textured surface.

As a postdoc in the Aizenberg group, Tak-Sing Wong, now an assistant professor at Pennsylvania State University in the US, developed surfaces based on the pitcher plant principle, called slippery liquid-infused porous surfaces (Slips). ‘You start with a textured or porous sponge-like surface and then we put on a lubricating fluid that has a strong chemical affinity to the underlying textured [surface], and with this combination a Slips can repel anything that’s immiscible with the lubricant,’ he says. ‘If you want to repel oil-based fluid then you can engineer the lubricant to either be aqueous or you can use a perfluorinated fluid which is immiscible with a water and an oil phase. Based on these design criteria you can explore all kinds of liquids as a lubricant.’
The lubricant, typically 100nm to a few micrometres in depth, is held in place by capillary forces but it must have a high chemical affinity to the underlying material, otherwise the fluid you want to repel will displace it. ‘In principle you can use any kind of material as long as you can find a way to texture it,’ says Wong. He has used porous epoxy-resins with 300nm diameter and 5μm high nanopores.8
Wong has been investigating how lubricated surfaces could also be useful for water collection, in parts of the world where there are water shortages. He found that a hydrophobic Slips did not provide the most efficient way to harvest water vapour or fog as it limited water drop nucleation. And simply using a hierarchical textured surface led to droplets being pinned as with the rose petal, rather than rolling off to be collected.9
Instead he and his team came up with a solution that takes inspiration from the pitcher plant and the rice plant, which they’ve called a slippery rough surface (SRS). To prevent water pooling, rice leaves have a hierarchical surface, with defined grooves forming a corrugated structure which forces water droplets to roll off in a perpendicular direction to these grooves. Wong and his team created silicon wafers with this structure. The grooved surface was itself modified with a second hierarchical texture and coated with silanes to enhance the chemical affinity with the lubricant. Only this innermost structure is coated with a 1μm hydrophilic silicone based oil lubricant.
‘Not only can it now attract the water vapour or water droplets from the air, but once the water is in contact with the surface it can slide away easily,’ explains Wong. A set of competing effects all contribute to a surface that repels liquids regardless of how they wet the surface. The hydrophilic lubricant helps water droplets nucleate and the rice leaf ridges then allow drops to roll off. Wong says current lab test have shown the system can collect roughly 500mg of water per cm2 in an hour: ‘close to 10 times more water than the typical fog-harvesting material’, he adds.
Nature has inspired diverse solutions to creating superhydrophobic and in some cases superoleophobic surfaces, on a range of materials. These solutions come from complex surface structures in addition to surface chemistry. But is this enough to replace the fluorinated polymers we currently use? Banerjee’s zinc oxid-coated mesh still uses an outer fluorinated layer to create superomniphobic behaviour. ‘I haven’t seen anything that works quite as well,’ he admits. ‘We did avoid using [bulk] fluoropolymers, instead using essentially monolayers.’ Werner thinks that in the future the right nanomorphology could replace fluorinated polymers for more environmentally friendly protective surfaces. His synthetic springtail skin is able to out-perform such surfaces. ‘They actually repel non-polar liquids and heavily contaminated aqueous solutions with very low surface tensions.’
But it’s probably not time to forget the chemistry altogether when designing materials. In investigating the springtail skin further Werner says the structured skin is not the whole story. ‘It turns out that this is maybe only the first line of defence of the organisms against bioadhesion,’ he explains. Surface chemistry and biology also play a role. A lipid-rich outer layer contains organic compounds known to provide anti-bacterial properties by minimising protein adhesion.10 ‘This also has something to offer us for biomimicry applications,’ Werner suggests.
Clearly we still have a lot to learn from nature. ‘I think we are just cracking the surface,’ agrees Banerjee.
Rachel Brazil is a science writer based in London, UK
References
1 W Barthlott, M Mail and C Neinhuis, Philos. Trans. R. Soc. A, 2016, 374, 20160191 (DOI: 10.1098/rsta.2016.0191)
2 RD Mukhopadhyay, B Vedhanarayanan and A Ajayaghosh, Angew. Chem. Int. Ed., 2017, 56, 16018 (DOI: 10.1002/anie.201709463)
3 R Hensel, C Neinhuis and C Werner, Chem. Soc. Rev., 2016, 45, 323 (DOI: 10.1039/c5cs00438a)
4 R Hensel et al, NPG Asia Materials, 2013, 5, e37 (DOI: 10.1038/am.2012.66)
5 O Tricinci et al, ACS Appl. Mater. Interfaces, 2015, 7, 25560 (DOI: 10.1021/acsami.5b07722)
6 T E O’Loughlin et al in Encyclopedia of Inorganic and Bioinorganic Chemistry, Wiley, 2017 (DOI: 10.1002/9781119951438.eibc2493)
7 T E O’Loughlin et al, Adv. Eng. Mater., 2017, 19, 1600808 (DOI: 10.1002/adem.201600808)
8 T-S Wong et al, Nature, 2011, 477, 443 (DOI: 10.1038/nature10447)
9 X Dai et al, Sci. Adv., 2018, 4, eaaq0919 (DOI: 10.1126/sciadv.aaq0919)
10 J Nickerl et al, J. R. Soc. Interface, 2014, 11, 20140619 (DOI: 10.1098/rsif.2014.0619)












1 Reader's comment