James Mitchell Crow explains how an unexpected discovery in main group chemistry inspired two decades of chemical creativity, from carbon dioxide reduction to fluorocarbon recycling, offering sustainable alternatives to precious metal catalysis
- A serendipitous discovery in Douglas Stephan’s lab 20 years ago revealed that sterically hindered Lewis acids and bases – frustrated Lewis pairs (FLPs) – can cooperatively activate small molecules, beginning with the first non‑metal reversible hydrogen activation.
- FLP chemistry has since expanded far beyond hydrogenation, enabling activation of diverse substrates including olefins, alkynes, carbon dioxide, carbon disulfide and even allowing asymmetric catalysis, radical pathways (via frustrated radical pairs) and new routes in organic synthesis.
- Researchers are applying FLPs to selective carbon–fluorine bond activation, enabling fluorocarbon upcycling, controlled monodefluorination and pharmaceutical radiolabelling with fluorine‑18 for PET imaging.
- Current efforts focus on rational FLP design to boost catalytic activity, improve moisture tolerance and expand applications, including CO₂ reduction, polymerisation catalysis and immobilising FLPs in MOFs for stability and recyclability.
This summary was generated by AI and checked by a human editor
At any given moment, how many reactions stirring away in flasks around the world are quietly veering off course, generating products nothing like what was anticipated? Even subtle-seeming changes in starting materials can send a textbook reaction careening in an unexpected direction. The deviously complex rulebook of chemical reactivity can seem to have been written by highly mischievous gods. But sometimes a mortal catches out the chemistry gods at their own game. One unexpected reaction result can trigger a flash of insight that unlocks a whole new chapter in the reactivity rulebook.
For Douglas Stephan at the University of Toronto in Canada, that moment of insight came 20 years ago this year, when a reaction that went awry inspired the concept of frustrated Lewis pairs (FLPs). Stephan and his team discovered that select pairs of bulked-up main group complexes, too sterically encumbered to react directly with each other, could cooperatively react with dihydrogen instead. The reaction was the first non-metal reversible hydrogen activation ever reported.
‘At the beginning, I thought FLP chemistry was going to be restricted to hydrogenation catalysis,’ Stephan says. But the team soon showed that FLPs could activate many other small molecules in similar fashion. ‘The more we learned, the more obvious it became that this was a paradigm for reactivity that needed broad exploration.’
Two decades later, the implications and applications of Stephan’s discovery continue to ripple outward. FLP chemistry now touches asymmetric catalysis, polymer synthesis, pharmaceutical radiolabelling, environmental remediation and beyond, as a growing contingent of chemists apply the concept in ways Stephan never anticipated.
From Lewis theory to frustrated pairs
Lewis pairs are a classic concept in main group chemistry, named following the expanded theory of acids and bases put forward by Gilbert Lewis in 1923. He defined bases as any substance that can donate an electron pair, and acids as any electron pair acceptor.
Lewis acids and bases generally make contented flaskfellows. A Lewis acidic borane, with its vacant electron orbital, will readily combine with a lone-pair-bearing Lewis base such as an amine or phosphine, for example, to form a stable adduct bound by a dative bond. But there has long been hints of unusual behaviour between bulkier Lewis acids and bases.
‘There’s been observations as far back as the 1940s of systems that didn’t behave the way you might expect donors and acceptors to behave,’ Stephan says. H. C. Brown, for example, showed that the sterically hindered amine 2,6-lutidine doesn’t form the expected adduct with trimethylboron. ‘He rationalised that it was too bulky to form a dative bond, and left it at that, never exploring what that might offer,’ Stephan says.
Even in Stephan’s own lab, examples of what we’d now recognise as FLP chemistry slipped by. It was only when a student accidently made an FLP that the team realised was reversibly activating hydrogen that the concept began to crystalise.
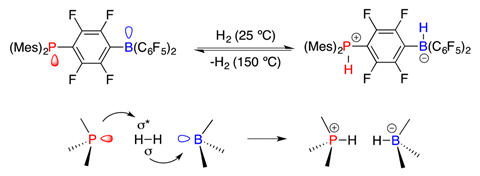
At the time, the group was focused on organometallic chemistry. ‘The original intent was to make an anionic phosphine with a borane attached, which would be an interesting ligand,’ Stephan says. The team got so sidetracked by the product they made, they didn’t return to the ligand idea for more than a decade.
That unexpected product, [mes2P+H(C6F4)B–H(C6F5)2], was an exotic zwitterion containing both a protic and a hydridic fragment. Heating it released hydrogen. But the resulting product could be converted back to the zwitterion by exposing it to hydrogen at room temperature and pressure. Simpler Lewis pairs, also too bulky to react directly, formed the equivalent salts when exposed to hydrogen, the team showed.
‘Once we realised that we could activate hydrogen, we saw that we should be able to activate all kinds of small molecules,’ Stephan says. ‘The next step was to do it with olefins, and that’s when we coined the phrase frustrated Lewis pairs.’
FLPs’ potential as sustainable catalysts made of earth-abundant elements, as alternatives to expensive precious metals, was immediately clear. The first catalytic examples Stephan demonstrated were hydrogenations of imines, where the imine also acted as the FLP base. Adding hydrogen and catalytic B(C6F5)3 as the Lewis acid, the activated hydrogen would initially protonate the imine. Hydride transfer from the borate to the iminium carbon completed the reduction and regenerated the catalyst. The team used this chemistry to reduce imine precursors for herbicides, antidepressants and anticancer drugs. FLP hydrogenation chemistry was gradually broadened to include alkene, alkyne and carbonyl reductions.
FLP catalysis was also expanded to the activation of many other small molecules ‘Besides hydrogen, we’ve looked at activating olefins, alkynes, carbon dioxide, carbon disulfide…’ Stephan says. The one small molecule that has so far resisted FLP activation is the ultra-stable, triply bonded nitrogen molecule.
Some of the latest advances in this space are chiral FLPs for metal-free asymmetric catalysis, including hydrogenation and hydrosilylation. ‘The bulk of that work comes from Haifeng Du at the Chinese Academy of Sciences’ Institute of Chemistry in Beijing, who has extended FLPs to highly enantioselective catalysis, which is pretty impressive,’ Stephan says.
The radical alternative
FLP small molecule activation was originally conceived as a purely heterolytic process with electrons remaining paired. But some FLP reactions have shown hints of radical behaviour. Researchers realised that single electron transfer from frustrated Lewis base to acid could generate a frustrated radical pair (FRP). FRPs can activate substrates via homolytic bond cleavage, enabling access into radical chemistry.
‘We came upon FRP chemistry by accident after we found we were getting radicals in a reaction,’ says Rebecca Melen from Cardiff University, UK, who focuses on FLPs for organic synthesis. ‘We now design FRPs to use in single electron transfer reactions, to open a new field of reactivity in organic chemistry.’
Even the classic B(C6F5)3/P(mes)3 FLP can form FRPs with aryl esters, leading to carbon–carbon bond formation, Melen has shown. ‘We are interested in the reactivity, but also in understanding how we’re generating the radicals,’ Melen says. ‘It’s a very complex system where potentially we’re getting radicals by electron transfer from the Lewis base to the Lewis acid-activated substrate.’
Expanding the catalytic toolkit
FLPs offer more than just metal-free alternative catalysts for known reactions. They often give access to alternative reaction selectivity. One example is FLP-mediated carbon–fluorine bond activation, says Rowan Young, who has focused on this chemistry at the University of Queensland in Australia.
Stephan published the first examples of C–F bond activation. ‘Our closest competition in the area probably is Doug, but he’s very friendly about that!’ Young says. Young’s group has focused on using FLPs to upcycle small molecule fluorocarbons at the end of their life.
‘PFAS has a lot of publicity at the moment, but the fluorocarbons produced on megaton scale are things like refrigerants, which have very high global warming potential,’ Young says. ‘We’re aiming to develop ways to reuse these things for economic benefit, rather than just destroying them.’

One high volume use could be to tap used fluorocarbons as a source of fluorine gas, which has several industrial scale applications. Young has also investigated fluorocarbon recycling for high value pharmaceutical or agrochemical applications, which comes with a chemical challenge that FLPs have proven ideally suited to solve.
‘The challenge is to functionalise a polyfluorocarbon C–F bond in a controlled manner,’ Young says. The carbon–fluorine bond’s strength stems from fluorine’s high electronegativity. The more fluorine atoms attached to a carbon atom, all drawing electron density from the carbon, the more polarised and stronger each carbon–fluorine bond will be.
‘If you’re trying to selectively functionalise one fluorine bond of a CF3 group, replacing that first carbon–fluorine bond makes the remaining carbon fluorine bonds weaker, resulting in uncontrolled over-defluorination,’ Young says.
But with FLP carbon–fluorine bond activation, the selectivity is switched compared to traditional methods. As the Lewis acid abstracts the first fluoride from a CF3 group, it generates a CF2 cation that is captured by the Lewis base. ‘Abstracting a second fluoride from something which is already cationic becomes much more difficult,’ Young says. ‘The second and third C–F activations get progressively harder, not easier, so we get monoselective conversion into our desired FLP activated product.’
Different Lewis bases enable access to different final products. ‘If we want to replace the C–F bond with an aryl group, we’ll use a thioether as our Lewis base,’ Young says. The captured cationic fragment will be a sulfonium. ‘There’s very rich palladium coupling chemistry around sulfonium salts, and we can utilise that chemistry to install an aryl group,’ he says.
Alternatively, FLPs can selectively pluck a single fluorine from a pharmaceutical with a CF3 group, to replace it with radioactive fluorine-18. The approach enables late-stage pharmaceutical radiolabelling for PET medical imaging studies, a valuable advance given fluorine-18’s brief 110-minute half-life.
Early steps in industry
For FLP small molecule activation, some of the latest advances have come in catalytic carbon dioxide reduction, potentially offering a way to recycle captured carbon emissions into valuable products. Following the FLP-mediated catalytic conversion of carbon dioxide into formate (HCO2−), Stephan and his collaborators developed a metal-free system to reduce it all the way to methane. The team combined 2,6-lutidine and B(C6F5)3 as the FLP, using hydrogen to reduce the FLP-activated CO2 molecule. The trick was to add a silylhalide as an oxophile, helping to free the borane for subsequent catalytic cycles.
These metal-free methods for small molecule activation are often not as fast or efficient as transition metal catalysed efforts, Stephan acknowledges. ‘I’d argue that transition metal chemistry had a 60- or 70-year head start,’ he adds. FLPs are yet to jump from academic advance to commercial use, but some of the first applications could be in polymerisation catalysis, Stephan says.
Dow Silicones is one company that has been exploring this chemistry, including FLPs as heat-triggered catalysts for curing siloxanes used in coatings, adhesives and sealants. ‘Dow told me that some of their FLP work was getting close to commercialisation, though they didn’t share any details,’ Stephan says.
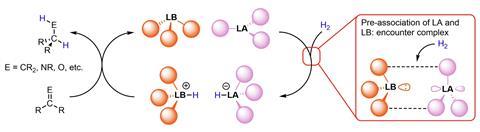
The way to catalyse broader FLP commercial uptake is using rational design, the same approach that enabled modern industrial transition metal catalysis, argues Andrew Jupp at the University of Birmingham, UK. ‘FLPs haven’t been adopted by industry, despite their green credentials, because they’re just not active enough yet,’ he says.
Designing FLPs that pair more strongly could be the key to boosting catalytic activity, Jupp says. ‘FLPs are held together by really weak interactions, so they can have a host of different conformations in solution – they’re not strongly held together in their catalytic form,’ he says.
Weak interactions also make catalytically active FLP encounter complexes hard to study experimentally. But some colourful FLPs – including Stephan’s prototypical P(mes)3 and B(C6F5)3 combination – can be probed, Jupp has shown.
‘Mixing the colourless phosphine and borane, you get a beautiful magenta colour,’ Jupp says. The colour arises from single electron transfer driven by light absorption, Jupp explains. ‘For this transfer to take place, there needs to be a path for that single electron to hop from the Lewis base to the acid – which can only occur when the pair are in the catalytically active conformation,’ he says.
The team studied coloured FLP encounter complexes using a UV–vis spectroscopy technique borrowed from supramolecular chemistry. ‘Analysing the intensity of the charge transfer band, we can get key thermodynamic information on how the FLP interacts,’ Jupp says.
Jupp’s results suggest there is plenty of scope for FLP catalyst improvement. ‘Our numbers correlate nicely with what’s predicted computationally – which is that the stabilising interactions holding the encounter complex together are almost perfectly cancelled out by entropy, so the favourability of them coming together in solution is about zero,’ Jupp says. Only about 1% of the FLP is in the catalytically active conformation at any one time, the numbers suggest. ‘We want to come up with systems where we can increase that number significantly, to make these catalysts much more active.’
The team is now using UV–vis spectroscopy to systematically analyse encounter complex formation in a range of FLP combinations. ‘Those findings will hopefully give us some design principles to develop new and improved ones,’ Jupp says.
Jupp is also targeting ways to make FLPs more user-friendly. ‘Something that’s plagued FLPs is that they’re very good at activating small molecules – including water – so you have to use glove boxes and Schlenk lines to exclude water when setting up these reactions.’
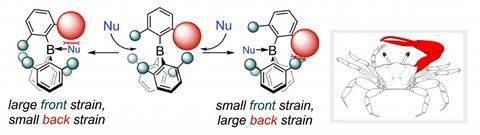
One approach to moisture-tolerant FLP hydrogenation catalysts, explored by Tibor Soós at the Institute of Organic Chemistry in Budapest, Hungary, has been to modify the borane to control the size of the reactive pocket within the FLP encounter complex. The pocket is tuned so that hydrogen fits, but water doesn’t. The team’s latest designs, which extend the steric FLP design concept to selective ester reduction to aldehydes, are dubbed fiddler crab boranes for their single oversized side arm.
Jupp is targeting more inherently water-tolerant Lewis acids than boranes. ‘We’re looking at indium – from the same group as boron – which can interact with water but tends to do so reversibly.’
FLP behaviour has already been expanded from main group complexes to most of the periodic table. FLPs with transition metal complexes as the Lewis acid or base, or rare earths or even alkali metals as the Lewis acid, have all been demonstrated.
Two decades on from Stephan’s original discovery, there’s no sign that the concept’s expansion into new areas of chemistry is slowing. ‘There’s a lot of work now going on to putting FLPs in metal-organic frameworks [MOFs], which means you can make FLP catalysts more selective, stable and recyclable,’ Stephan says. ‘It’s fantastic to see people take your idea and run with it in completely different directions.’
James Mitchell Crow is a science writer based in Melbourne, Australia


















No comments yet