Why do so many biological molecules exist in just one chirality – and how did it emerge? Rachel Brazil reflects on life’s strange asymmetry

Life exists in single-handed forms. Asymmetric carbon atoms cause chirality in molecules – the possibility of two mirror image forms. Almost all chiral molecules in living organisms are found in just one form: sugars are exclusively right-handed, amino acids left-handed and DNA coils into right-handed helices. Some think that this single-handedness, or homochirality, was a prerequisite for the formation of the replicating molecules that led to all life. But the big question is how did our single-handed world develop from a primordial chemical soup that was both left- and right-handed? Often described as ‘breaking the mirror’, the problem may seem trivial at first glance, but chemists still do not understand how it happened.
The presence of carbon atoms with four chemically distinct substituents leads to chiral molecules, with non-superimposable mirror image forms known as stereoisomers. In any chemical synthesis, without intervention, an almost equal or racemic mixture of left- and right-handed molecules will form. So where did our homochiral world come from?
Donna Blackmond from the Scripps Research Institute in California, US, has been tackling the question. She says there is a rationale for why homochirality might offer a survival advantage. Chirality is important for creating complex systems with varied forms of molecular recognition and selectivity – both of which are crucial in life. Blackmond says when she gives talks to children, she uses a handshake to explain these concepts. ‘We shake hands and then I say “Let me try this with my left hand and your right hand” – and that doesn’t work very well. Left and right doesn’t make the same handshake or give the same message.’
No homochirality, no life
The first experimental confirmation that key organic molecules of life could be created from a primordial soup, perhaps 4 billion years ago, came in Stanley Miller’s classic 1953 experiment1 where an electric spark, simulating lightning, led to the creation of organic molecules from water, methane, ammonia and hydrogen. He managed to produce 11 of the 20 naturally occurring amino acids – but all were produced equally in the left- and right-handed forms.
Today, the RNA world hypothesis is largely accepted: self-replicating RNA molecules were the precursor to DNA as the mode of inheritance, as they are simpler to synthesise. In 2009, John Sutherland, then at the University of Manchester in the UK, showed that chemical units that can be polymerised to make RNA can be synthesised from plausibly prebiotic starting molecules: cyanamide, cyanoacetylene, glycolaldehyde, glyceraldehyde and inorganic phosphate.2 But this still requires using only the right-handed version of the sugar glyceraldehyde to create the correct biological products.
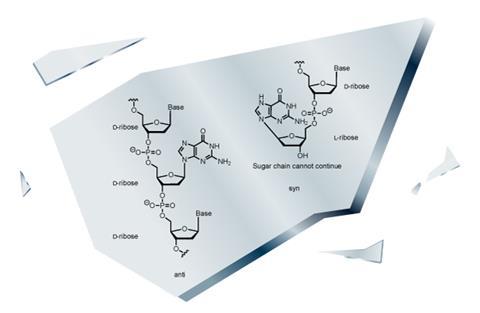
With both stereoisomers present, RNA cannot be polymerised. According to Blackmond, some people say ‘no homochirality, no life’ and so far that seems to be true. The big sticking point is what is known as enantiomeric cross-inhibition,3 or what Gerald Joyce jokingly calls biology’s ‘original syn’.
Joyce, also at Scripps, coined this term over 20 years ago to describe why only single-handed chemistry can build biological polymers like RNA, and why the opposite handed molecules block assembly. ‘It turns out if you have nothing but one hand, the chemical copying mechanism works very nicely,’ he says, but things go wrong when there are building blocks of both chirality. ‘They basically poison each other’s polymerisation.’
The cross-inhibition occurs because nucleic acids start polymerisation by matching base pairs. Incoming monomers add onto the backbone with each sugar and base configured anti to each other. But Joyce explains that if a monomer with a sugar of opposite handedness is present, then for it to both base-pair and bond to the backbone, it must add with its base in the syn configuration, which creates a conformation that blocks further polymerisation. ‘Then the show is over,’ says Joyce. ‘It really is the original syn that goes all the way back and has to be overcome somehow – not through divine intervention, but through chemistry.’
So given a racemic prebiotic world, how did we get RNA? ‘People used to think there was this kind of chicken and egg problem: you can’t break the mirror until you have biology, but you need to break the mirror to get biology started.’ The search is now on for a method that allows symmetry breaking – creating homochiral products from non-chiral starting materials.
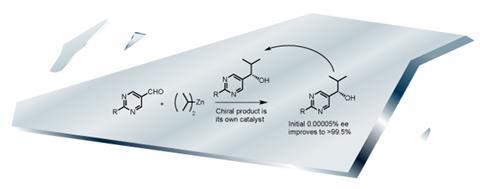
A theoretical model for symmetry breaking was proposed in the 1950s by British physicist Charles Frank, who suggested that in autocatalytic reactions, where the products are catalysts for their own formation, any small random asymmetry in the products will be amplified and will provide a way of enhancing one hand over another. A reaction confirming this idea was only found in 1995 by Japanese chemist Kenso Soai.4 His autocatalytic alkylation of pyrimidyl aldehydes with dialkylzincs is catalysed by its chiral alcohol product. Given a very small excess of the product in one chirality, the eventual yield of the reaction will have much more of that form than its mirror image – as high as 91% excess. ‘It gave experimental proof of concept,’ Blackmond says, but points out the one big flaw: ‘The actual chemistry isn’t at all prebiotically relevant.’
Crystal-clear chirality
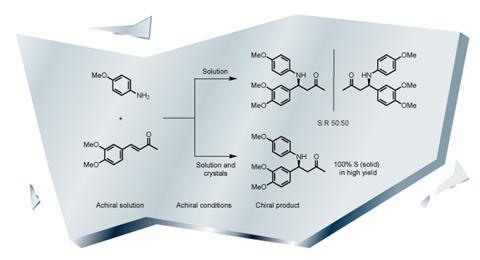
Another approach is to look at the effects of crystallisation. The observation that crystals can be homochiral goes back to Louis Pasteur, who noticed in 1848 that chemically synthesised ammonium sodium tartrate formed two mirror-image types of crystals that he could sort into two piles. Elias Vlieg at Radboud University in the Netherlands has worked with Blackmond and others to develop homochirality via this route. In 2014, he published a method to make a homochiral amine by precipitating and repeatedly crystallising the product.
‘From my perspective this is almost the simplest mechanism of getting single chirality,’ Vlieg says. ‘In the solid state you can really have amplification of symmetry breaking and go from 0 to 100% chirality.’ This is because of the Ostwald ripening process, where over time small crystals tend to spontaneously redeposit into larger ones, which are thermodynamically more favourable. It’s something you often see when clearing out old laboratory cupboards, Vlieg says. ‘Quite often you find a surprise: some beautiful crystals that have been Ostwald ripening forever – this can take a very long time but eventually you end up with a single crystal.’
The natural hand of the amino acid sequesters the unnatural hand of the sugar, that’s how the amplification occurs
Vlieg and his colleagues reacted achiral starting materials that produce an equal mixture of left- and right-handed amine products, but this changes with crystallisation.5 They used a process called Viedma ripening, a phenomenon first reported in 2005 by geologist Cristobal Viedma.6 By continually grinding the crystals in solution for a couple of days you can speed up the Ostwald ripening. ‘You can in a straightforward single pot get 100% homochirality,’ says Vlieg. Temperature cycling seems to have a similar effect, but the method only works for molecules where the left- and right-handed versions crystallise separately, and this is not always the case. Vlieg says they are now trying to do the exact same process with an amino acid, but it is proving more difficult.
Blackmond contends that it’s not enough to find a method that creates a single chiral product from achiral starting materials – the reaction has got to provide a way of doing this with the sorts of molecules that might have been present in the prebiotic world. In 2011, she found a way to take Sutherland’s route to RNA but make molecules of a single chirality. Her route to making RNA precursor molecules from racemic glyceraldehyde and 2-aminooxazole still requires a chiral amino acid.7 Her ‘kinetic resolution’ method works by creating a reaction where one form of a molecule can react faster than the other. ‘You need something chiral to interact with them differently and we developed that,’ Blackmond says.
Her reaction worked best with the left-handed l-amino acid proline. Using just 1% excess of the l form, it was possible to produce a product of 100% chiral purity. The process works because the l-amino acid reacts at different rates with the two stereoisomers of glyceraldehyde. The l-sugar reacts more quickly with the l-amino acid and, on reacting with the oxazole, produces a homochiral three-component product. A similar preference was seen with the right-handed d-amino acid and d-sugar.
‘It’s an amazing reaction,’ says Blackmond. ‘It turns out that the natural hand of the amino acid sequesters the unnatural hand of the sugar, and vice versa, and that’s how the amplification occurs. So the sugar can be used to amplify the amino acid enantiomeric excess, or the amino acid can be used to amplify the sugars. Truly chicken or egg … or egg or chicken!’ Blackmond suggests that the enriched amino acid could have formed initially via a crystallisation process. ‘It could have been all of these things contributing a little bit over a long time.’
Breaking the mirror
University College London chemist Matthew Powner worked with Sutherland in synthesising prebiotically plausible RNA precursors and continues to develop synthetic strategies to explain early chemical evolution. He says understanding homochirality is still completely unresolved. ‘There are nice ideas out there, but none of them have been adequately demonstrated.’ We also don’t know the stage at which symmetry broke. Did it happen once or many times in many little ponds? Was it a gradual process with increased enrichment developing over a number of chemical steps? Powner says one fundamental question is the relationship between the l-amino acids and d-sugars found in biology – are their conformations just an accident, or was it caused by some fundamental chemical reaction that transferred chirality from one to the other?
Maybe the mirror didn’t break – instead of one hand fighting the other, maybe one made the other
‘Chirality is not absolute in biology,’ Powner points out. For example, the monosaccharide l-arabinose is found in plant biopolymers such as pectin and hemicellulose from plant cell walls. Single-celled archaea use l-glycerol to make their phospholipid membrane, which is the opposite chirality to the d-glycerol used to build bacterial and eukaryotic membranes. This may mean that homochirality wasn’t always the case. ‘Maybe that’s part of the evolutionary arms race – starting to use something that other things don’t recognise, to give you an evolutionary advantage,’ Powner says.
The possibility that two types of chirality existed at the early stages of chemical evolution has also arisen from recent work by Joyce.8 He showed that it is possible to create a ribozyme – an RNA enzyme – that can replicate a ‘cross-chiral’ mirror image of itself without the help of any proteins or other cellular components. ‘No one had ever seen an enzyme in nature that works on the opposite hand – so we just wondered if it was possible,’ Joyce explains. The ribozyme was created by a complex chemical test-tube evolution process. Right-handed RNA molecules that were most successful at joining mirror image left-handed RNA strands were selected. This process was repeated multiple times to amplify the success of the copying mechanism, finally creating an 83-nucleotide ribozyme.
Joyce found that the ribozyme works with both left- and right-handed RNA – getting over the ‘original syn’ problem. Could this explain early chemical evolution? ‘Maybe you didn’t break the mirror and, instead of one hand fighting the other, maybe one hand makes the other,’ says Joyce. But at some point a homochiral chemistry did arise. Joyce suggests some evolutionary innovation at the beginnings of polypeptide synthesis might have led to the right-handed RNA chirality predominating.
A lucky draw
Another curiosity is why one side of the mirror predominated over the other. Is there a reason why biology has right-handed sugars and left-handed amino acids rather than everything being the other way around? Could we just as likely be living in a world made from a mirror-image of the biology we have today? ‘There is no real reason why one should be preferred over the other,’ says Vlieg. ‘There could be a planet somewhere out there which would have the exact opposite chirality.’
Somewhere there was a lucky coin flip that gave the D-sugar a big advantage
‘It’s just the luck of the draw,’ Blackmond agrees. Some have suggested that exposure to polarised radiation caused the imbalance. ‘The universe is not perfectly symmetric: in beta decay there is a slight asymmetry and the spin of the electron is slightly asymmetric,’ Joyce explains. But these effects are too small to explain the chemistry when other, much greater, energy differences are at play. ‘To a chemist this makes no sense – that slight physical asymmetry is simply swamped; and to a biologist it makes even less sense because biologists are used to thinking about coin flips – somewhere there was a lucky coin flip that gave the d-sugar-containing version a big advantage.’
Joyce says the excitement now is to try to make different forms of life to better understand the origin of homochirality. ‘Where the field is headed is to actually make a synthetic life form – not taking our lifeform and cobbling the pieces together in a different way, that’s what is usually meant by “synthetic biology” – but I mean a new synthesis of a truly different biology.’ Joyce says that since the publication of his cross-chiral RNA ribozyme in 2014, his team has evolved a better-functioning version. Geneticist George Church at Harvard University in the US has been trying to create a complete mirror-image cell.
If the coin had flipped the other way, perhaps we might be living in a mirror-image world, with a biology made from l-sugars and d-amino acids. Would we be able to tell the difference? Joyce says this is a purely theoretical question: ‘Even if you started with the same hand again, we’d end up in a different place.’ But if the coins all flipped in exactly the opposite way and all chirality was reversed, he says there is no reason why everything wouldn’t seem the same. Joyce says that in a recent lecture at the University of California, Berkeley, he showed a picture of the campus clock tower, but in mirror-image form to see if anyone would notice the difference. ‘It’s amazing how hard it is for people to know if they are looking at the regular thing or the mirror image. In that case you look at the clock face and the nine is on the right and the three is on the left and yet nobody noticed it. If all the coins flipped the same way everything would be the same except the nines would be on the right and the threes would be on the left!’
Rachel Brazil is a science writer based in London, UK
References
1 S L Miller, Science, 1953, 117, 528 (DOI: 10.1126/science.117.3046.528)
2 M W Powner, B Gerland and J D Sutherland, Nature, 2009, 459, 239 (DOI: 10.1038/nature08013)
3 G F Joyce et al, Proc. Natl Acad. Sci. USA, 1987, 84, 4398
4 K Soai et al, Nature, 1995, 378, 767 (DOI: 10.1038/378767a0)
5 R R E Steendam et al, Nat. Commun., 2014, 5, 5543 (DOI: 10.1038/ncomms6543)
6 C Viedma, Phys. Rev. Lett., 2005, 94, 65504 (DOI: 10.1103/PhysRevLett.94.065504)
7 J E Hein, E Tse and D Blackmond, Nat. Chem., 2011, 3, 704 (DOI: 10.1038/nchem.1108)
8 J T Sczepanski and G F Joyce, Nature, 2014, 515, 440 (DOI: 10.1038/nature13900)











1 Reader's comment