Dinoflagellates create some of nature’s most spectacular light shows, yet their bioluminescent chemistry remains poorly understood. Rachel Brazil explores how researchers are unravelling the unique reactions behind the sea sparkle phenomenon and what it might reveal about evolution and future technologies
- Dinoflagellates create spectacular bioluminescent displays such as Puerto Rico’s Mosquito Bay, yet the chemistry behind their blue–green flashes remains one of the least understood bioluminescent systems.
- Their light production is triggered by mechanical stimulation and involves a rapid signalling cascade inside specialised organelles called scintillons, where pH changes release luciferin to react with luciferase.
- Researchers are still debating the evolutionary origin and exact reaction mechanism, with uncertainties about the true structure of dinoflagellate oxyluciferin and whether the process mirrors other bioluminescent chemistries.
- New modelling studies and biochemical investigations aim to clarify the reaction pathway and uncover additional proteins involved, while bioluminescence continues to inspire novel technologies and underscores the need to protect fragile ecosystems like Mosquito Bay.
This summary was generated by AI and checked by a human editor
The best place to see the sea sparkle is a shallow, sheltered bay on the south coast of the tiny island of Vieques, located off the coast of Puerto Rico. Luckily for me, Mosquito Bay is not named after an infestation, but for an 1820s pirate ship that used the bay to hide. Last April I took my own trip there, to experience the brightest bioluminescent bay in the world. To get the most intense display, you kayak to the middle of the bay on a moonless, pitch-black night.
You don’t see a constant glow, but as each paddle enters the water it causes an electric blue–green explosion of light. Let a handful of the water run through your fingers and it twinkles like Peter Pan’s magical fairy dust. The phenomenon is caused by millions of microscopic Pyrodinium bahamense, a species of the group of single-celled plankton known as dinoflagellates.
Bioluminescence isn’t unique to dinoflagellates. ‘You have about 3500 different species which are bioluminescent,’ says organic chemist and bioluminescence researcher Stefan Schramm from HTW Dresden in Germany. ‘There are probably still many that have not been found,’ he adds. One of the best known systems is the firefly. In the 1940s, William McElroy from Johns Hopkins University in Baltimore, US, first extracted the firefly enzyme and substrate molecule involved in producing light. To do this he needed 15,000 fireflies, caught for him by local children. He also found the light-producing reaction was catalysed by ATP (adenosine triphosphate), the main energy currency of living cells. The molecules were subsequently commercialised as a kit for detecting ATP as a marker for living cells – one of the first products of the fledgling Sigma Chemical Company.
Why the sea sparkles
Of all the bioluminescent systems, the dinoflagellates are the least well understood and seem to stand out in their chemistry. ‘They started to study it back in 60s, but we’ve made quite slow progress in understanding how it all works,’ says ecologist Jan Janouskovec of Southampton University in the UK, who is working on the biosynthetic pathways that these plankton use to make their bioluminescence apparatus. Although dinoflagellates are in every ocean, they are often associated with tropical or sub-tropical waters. But Janouskovec says they are increasingly found around the UK – he collected his latest samples in Plymouth harbour and is hoping they will help him answer some of the remaining conundrums surrounding these flashing creatures.
Not all dinoflagellates are bioluminescent. ‘It’s scattered across the [evolutionary] tree,’ says Janouskovec. Those with the ability can vary greatly. Pyrodinium bahamense, found in Mosquito Bay, are thecate – meaning the cell is covered by a cellulose shell, creating numerous beautiful shapes. They are photosynthetic and can produce toxins, so when they accumulate in shellfish and are ingested they can be harmful to humans. Another species Noctiluca scintillans, often identified as ‘sea sparkle’ is athecate, which means it has no shell, commonly not able to photosynthesise and is usually non-toxic.
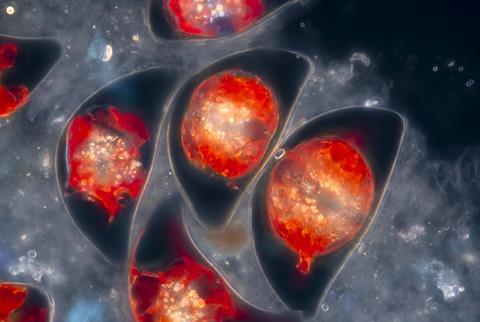
Even so, ‘there are times when it does bloom and become very abundant’, says marine biologist and dinoflagellate bioluminescence expert Michael Latz from the Scripps Institution of Oceanography at the University of California, San Diego, US. When this happens, it can be a danger to ecosystems. As with other aquatic blooms, they exhaust oxygen supplies which starves other wildlife and can devastate fisheries. This was recently seen along the southern coast of Australia. ‘We call them red tides, because during the day, the water is a discoloured reddish brownish,’ says Latz, but ‘at night, you get the beautiful bioluminescence in the waves as they break’.
Why do some species bioluminesce?
The purpose of dinoflagellates bioluminescence is another debated question. Uniquely compared to some other species, their bioluminescence is stimulated by mechanical force. So most researchers have interpreted it as an attempt to ward off predators when disturbed. But Latz thinks it might be more subtle than that. ‘It can actually act as a burglar alarm,‘ he says. His idea is that when a predator like a shrimp sets off the flashes, this provides a signal to secondary predators like squid who will come for the shrimp. ‘The bioluminescence would act like a minefield that makes other animals vulnerable,’ he says.
Chemist Schramm has a different suggestion for the origin of bioluminescence chemistry. He thinks these oxidation reactions could be a way for cells to get rid of damaging reactive oxygen species. ‘A current evolutionary hypothesis is that bioluminescence evolved just as a side product,’ he says. ‘When the Earth’s atmosphere became oxygen-rich, the organisms at that time couldn’t deal with the oxygen in a productive way, and needed to decompose it.’ A reaction that produced light was the perfect way to do this, and as with so much in biology, it was then repurposed for another task.
All dinoflagellate species use the same chemical reaction to produce light – which is unusually different from other bioluminescent systems. Confusingly for chemists, biologists have lumped all bioluminescing molecules (known as substrates) and their corresponding enzymes together, regardless of their chemical structure. ‘The enzyme is always called luciferase, the substrate is always called luciferin. The substrate gets oxidised within the reaction to an oxidised form, which is called oxyluciferin,’ explains Schramm. The oxidation usually goes through an intermediate peroxide which decomposes to the oxyluciferin via an excited state, which then emits a photon to return to its ground state.
Mechanical forces inside the scintillon
The bioluminescent chemistry in dinoflagellates occurs in an organelle called the scintillon, which sits inside the cell’s liquid-filled vacuoles. A mechanical force triggers a signalling cascade leading to the reaction. ‘It’s a complex signalling pathway,’ says Latz, but he and others have started to put the sequence together. It starts with a G protein, a common molecular switch that transmits signals from outside to inside cells, and then via a TRP (transient receptor potential) ion channel protein, which allows calcium ions to flow. Latz says the next step is still ‘a little mysterious’ but involves the depolarisation of the vacuole membrane which allows protons to flow into the scintillons via voltage gated ion channels, dropping the pH.
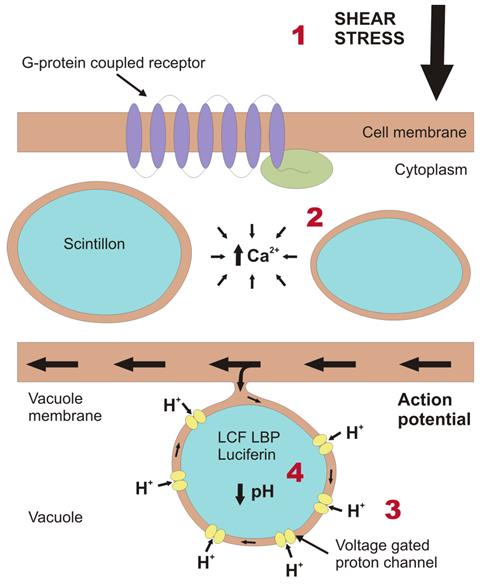
At higher pH the luciferin substrate is bound to the luciferin-binding protein, which protects against spontaneous oxidation, but when the pH drops below six the protein changes conformation and releases the luciferin molecule which can then bind to its luciferase counterpart where it is oxidised. ‘That whole process from stimulus to the flash of light only takes about 15ms, so it’s an extremely rapid signaling pathway, an almost instantaneous response, much faster than, for example, visual transduction,’ says Latz.
The chemical reaction of dinoflagellate bioluminescence still causes some head scratching and poses some interesting evolutionary questions. The luciferin is a non-cyclic tetrapyrrole with similarities to the porphyrin ring found in chlorophyll. Janouskovec is interested in that relationship. ‘People have started to speculate whether some of these organisms basically synthesise the luciferin from chlorophyll,’ he says.
The only problem with that idea is that not all bioluminescing dinoflagellates are able to photosynthesise. But Janouskovec says even they still have chloroplasts – the organelles where photosynthesis takes place – and use them to host other metabolic pathways. He wants to discover whether the synthesis of luciferin and luciferase in dinoflagellates shares a common synthetic pathway to chlorophyll. ‘From an evolutionary point of view, it’s fascinating when you find how things are repurposed.’ he says. His group is still working out the full luciferin synthetic pathway which they think includes 20 different enzymes.
Unravelling the bioluminescent mechanism
But the big conundrum is the mechanism of the bioluminescence reaction itself. The initial work was carried out in the 1970s and 80s by Princeton biochemist and 2008 Nobel laureate, Osamu Shimomura, who isolated and characterised green fluorescent protein (GFP) from jellyfish. GFP has become a crucial way to visualise biological processes in living cells by attaching it to genes or proteins which will then glow green under UV and blue light.
Schramm says there are some unusual anomalies surrounding the dinoflagellate reaction that are making him question some of the assumptions that have been made about the reaction. The most common bioluminescent reactions oxidise their luciferin leading to a decarboxylation and the loss of carbon dioxide. The reaction proceeds via a highly strained four-membered cyclic peroxide with an adjacent carbonyl group, known as a dioxetanone.

But there is as yet no agreement whether the dinoflagellate luciferin reacts, and whether the reaction is similar to the firefly and other bioluminescence chemistries. Of the more than 50 times bioluminescence has evolved independently, the chemistry always seems to converge to a mechanism of oxidation and decarboxylation, but the schemes suggested for the dinoflagellate progress without the loss of carbon dioxide. The oxidation of a methylene group to a ketone has been suggested based on the reaction product after the bioluminescence reaction.
One issue may be whether the correct oxyluciferin product was isolated in the 1980s. The published structure has not yet been experimentally verified to be the actual light emmter of the reaction. ‘I have some doubt,’ Schramm says. ‘Typically, the oxyluciferin should emit fluorescence at the same emission maximum of the bioluminescence reaction,’ but in contrast the molecule proposed as the dinoflagellate oxyluciferin does not fluoresce at all, which could imply this is not the molecule involved in the emission of light and merely a byproduct of the actual reaction.
Nevertheless, several groups have tried to model the reaction based on the published luciferin and oxyluciferin structures. The uncertainty around the reaction has made it an interesting case study for computational simulations, which have been able to tackle more complex problems in the last decade.
Computational conundrum
Theoretical chemist Filipp Furche at the University of California, Irvine, in the US is one who has recently taken up the challenge, together with Irvine chemistry colleague Jennifer Prescher, an expert on bioluminescence, and modelling expertise from collaborators at Los Alamos National Laboratory in New Mexico, US. They systematically investigated all possible arrangements of the molecule and luciferin intermediates including protonation states and stereochemistry and used modelling methods, including time-dependent density function theory (which models electron density changes over time), to examine the excited state dynamics.
They ran about 500 trajectories, each covering a femtosecond in the reaction profile. Furche says ‘the crucial part’ that the simulations can probe is at what stage and from what species the light is emitted. From their modelling they suggest that the luciferin is oxidised via a dioxetanol ring intermediate (a four membered ring with an OH group) at a carbon–carbon double bond adjacent to one of the pyrrole rings.
‘[The] point where both the O–H bond and the C–C bond are quite a bit stretched and only at this point is it possible to cross over into the electronically excited state and once it’s there, it’s trapped, and doesn’t easily go back.’ Furche explains the intermediate breaks up, creating two carbonyl-containing fragments and emitting a photon as visible blue–green light (~475nm) consistent with the observed dinoflagellate emission spectra.
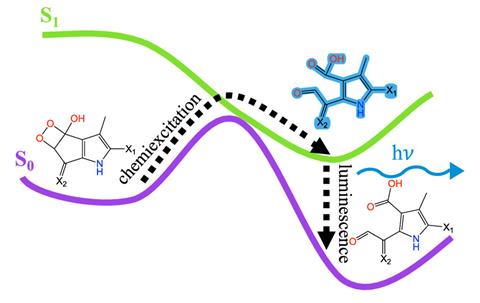
Their simulations predict an emission of 2.4eV in energy after a picosecond, which is close to the experimental emission at 2.6eV. ‘That’s the order of magnitude that one can expect from these simulations,’ says Furche.
The study found that only specific stereochemical arrangements and protonation states might be able to form an excited state. In nature this is likely controlled by the luciferase enzyme which will stabilise one stereoisomer and selectively block other ways the luciferin could react. But their modelling did not include the luciferase enzyme which Schramm says means, ‘the results remain purely theoretical’. Furche acknowledges the enzyme could have a very pronounced effect. ‘That’s something we are chewing hard on right now,’ he says. But adding the enzyme’s contribution to an already complex system would be very difficult.
Schramm is still not convinced that this modelling is looking at the right reaction. He thinks it’s likely that the proposed oxyluciferin product is actually another secondary oxidation product. ‘Dinoflagellates are either a very special case of bioluminescence, which I find bit strange seeing that other systems work via the formation of peroxide containing high energy intermediates and subsequent decarboxylation,, or this is not the correct oxyluciferin,’ he concludes.
There is clearly more work to be done to understand the bioluminescence chemistry of dinoflagellates. Plus, Janouskovec is keen to understand and identify other proteins that participate in the mechano-transduction and bioluminescence pathway inside the scintillons. ‘We know of two proteins [luciferase and the luciferin binding protein] and we now have [discovered] a third one, which is the pump that channels the protons inside. But you have a whole organelle which almost certainly has more protein components, and we just don’t know what they are.’
Latz says that will need more proteomic and genomic studies on dinoflagellates. ‘They have huge genomes, about 20 times the size of the human genome,’ he says, not because they have 20 times the number of genes, but because they unusually have no transcription factors, so are not able to switch genes on and off. Instead they have multiple gene copies, including 50–200 copies of the luciferase gene which is permanently transcribed – an alternative but apparently successful evolutionary strategy.
New chemistries and new applications
In addition to dinoflagellates, researchers are still searching for new luminescent chemistries. ‘You never know what tools they might offer in the future,’ says Latz. It’s challenging work because once you find an organism you need to be able to collect and culture enough of it to study the chemistry.
Furche says his group is now wading into ‘more difficult problems’ to understand other light emitting systems, together with Prescher who is designing an ingenious RNA tracking technology inside cells, based on bioluminescence. She is engineering RNA molecules that bind genetically encoded luciferase fragments via RNA-binding proteins. Only when the target RNA is present and correctly assembled will the luciferase fragments be brought together, reconstructing the enzyme. Then if the corresponding luciferin is added, light is emitted and the RNA can be detected. The beauty of the strategy is it avoids the interference or cell damage that can occur from exciting cell with external light sources.
We want to see if we can use them as a sustainable, biodegradable alternative to classical emitters in organic electronic applications
Schramm is also looking to further exploit bioluminescent chemistry. Nature has found a way to stabilise the reaction pathway leading to light emission and block the alternatives, he explains. ‘You see different structures that nature has optimised over the course of 200 million years of evolution to give very efficient light emission. For instance, with the firefly, you reach a bioluminescence quantum yield of about 42%, which is more efficient than every organic light emitting diode we can construct.’ Now he is trying to mimic these structural principles to engineer what he is calling BiOLEMs, biologically inspired organic light-emitting molecules. ‘We want to see if we can use them as a sustainable, biodegradable alternative to classical emitters in organic electronic applications.’
But to continue studying this phenomenon means preserving existing ecosystems, which takes us back to Mosquito Bay in Vieques. It has some unique factors that have sustained the highest concentrations of bioluminescent dinoflagellates anywhere. ‘The bay has a narrow opening to the sea, so the organisms that grow are retained,’ says Latz. ‘Another factor is that there are mangroves that fringe the shoreline.’ Their extensive roots are colonised by bacteria that produce B vitamins, which dinoflagellates need to thrive.
That isn’t a given, and such environments ‘are very much under threat’, says Latz. ‘They’re dynamic processes, and there’s no guarantee that they will last.’ He recalls that after hurricane Maria in 2017 the bay went ‘dark’ for weeks, due to freshwater influx and damage to the surrounding mangroves. It luckily started to recover three months later. But there are constant threats from chemical pollution and over-use by tourists.
Not all similar sites are protected, but Mosquito Bay was fortunately designated a natural reserve by the Legislative Assembly of Puerto Rico in 1989 and its use is carefully controlled. Protecting the bay means no engines or large boats and no insect repellent or swimming in the water for tourists, who can only visit with licensed tour guides. If their efforts are successful, future travellers will hopefully still be able to kayak into the bay on a moonless pitch-black night and experience the sea sparkling.
Rachel Brazil is a science writer based in London, UK


















No comments yet