After nine years of effort, chemists in the US have completed the first total synthesis of bacteriochlorophyll a, a molecule they describe as ‘one of the most challenging photosynthetic tetrapyrroles to synthesise’. By combining ‘battle‑tested and reliable’ methods, Jonathan Lindsey at North Carolina State University says his team’s approach opens the door to making a wide range of photosynthetic tetrapyrroles and exploring their function ‘in a way that’s not been possible previously’.
Photosynthetic tetrapyrroles are organic compounds made up of four pyrrole rings. Common examples are bacteriochlorophyll a and chlorophyll a, the main pigments in anoxygenic and oxygenic photosynthesis, respectively. While they share similar biosynthetic pathways, they operate in different organisms under varying environmental conditions, making them important for ecological diversity.
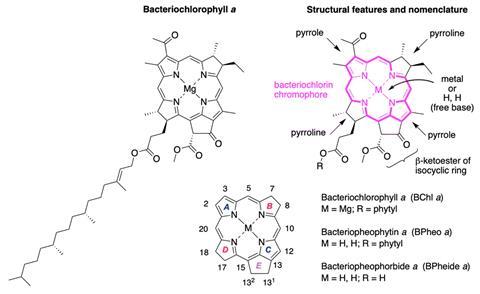
Despite their importance, chemists have largely neglected the chemical synthesis of these molecules. One reason is their daunting complexity. Bacteriochlorophyll a has four stereocentres, two in each pyrroline ring, at the edge of its bacteriochlorin chromophore. It also has an epimerisable β-ketoester embedded in an isocyclic ring, which makes it susceptible to oxidative dehydrogenation into chlorin or porphyrin derivatives.
Another reason dates back to 1960, when attempts to synthesise chlorophyll a set such a high bar that few were willing to follow.2 While that work did not synthesise chlorophyll a in a vial, it proposed an important pathway to get there. ‘It was so daunting and heroic what they had already accomplished,’ explains Lindsey. ‘I think after that, the field just became quiescent.’
Against this backdrop, Lindsey and his team set out to synthesise bacteriochlorophyll a, but there were challenges along the way. Firstly, the Covid-19 pandemic caused a global shortage of chemicals, so they had to revise some methods. Then, in February 2025, the project was rocked by the sudden death of team member Khiêm Châu Nguyễn from glioblastoma. The loss came as a profound shock, slowing the team’s scientific output while weighing heavily on them psychologically. Even after they overcame the experimental hurdles, validating the stereochemistry of the crystalline structures proved painstakingly slow. As a result, a project initially expected to take three to five years instead took closer to 10.
The method they have developed starts with an asymmetric Michael addition of a nitro-alkene and aliphatic aldehyde, such as propanal or butanal. This establishes the stereochemical configuration of substituents in precursors to the pyrroline rings, including bacteriopheophorbide a and bacteriopheophytin a. They then join two halves of the molecule with the isocyclic ring by a Knoevenagel condensation, followed by a double-ring closure involving Nazarov cyclisation and electrophilic aromatic substitution.
To synthesise the molecule ‘with all four stereocentres in the macrocycle, starting from readily available precursors, is an impressive achievement,’ comments Ana Moore, whose work at Arizona State University in the US focuses on synthesising analogues of photosynthetic reaction centres to better understand the light-driven and catalytic aspects of photosynthesis. ‘It is most impressive that the closing of the macrocycle with the two halves is accomplished with the formation of the isocyclic ring with the correct stereochemistry.’
Lindsey now plans to ‘refine the synthesis to speed it up, remove some of the wasteful steps and improve some of the yields.’ He also hopes to use it for other photosynthetic tetrapyrroles, and Moore agrees that ‘the design is so streamlined, it could certainly be adapted to synthesise other native photosynthetic pigments.’ She describes it as ‘all together a synthetic tour de force’.
References
1 DTM Chung et al, Chem. Sci., 2026, 17, 7454 (DOI: 10.1039/d5sc10233b)
2 RB Woodward et al, J. Am. Chem. Soc., 1960, 82, 3800 (DOI: 10.1021/ja01499a093)













No comments yet