Poly(ethylene glycol) is widely used in medicines and cosmetics but polydisperse mixtures complicate analysis and reproducibility. This article explores how monodisperse PEG with uniform chain lengths offers greater precision and control in drug development, improving both research and formulation outcomes.
Did you brush your teeth this morning? If so, you have already encountered poly(ethylene glycol) (PEG). It is one of the most ubiquitous polymers in personal care and pharmaceutical products. PEG serves as a surfactant and an emulsifier in cosmetics, while in medicine, it improves drug solubility and extends circulation time in the bloodstream.
Typically, an ingredient list or drug leaflet lists PEG with an average molecular weight. This means that polydisperse PEG is used, which consists of a mixture of varying chain lengths and often contains residual impurities. While polydispersity is acceptable for bulk ingredients like shampoos and body lotions, advanced pharmaceutical applications require precision that only monodisperse PEG can provide. By containing single-length molecules with an absolute molecular weight rather than an average, monodisperse PEG streamlines R&D and ensures clear analysis of the final formulations.
Challenges in drug development with polydisperse PEG
Polymers are inherently polydisperse materials, which is a natural consequence of the statistical nature of polymerisation.1 PEG is a member of the polyether family and is typically synthesised via the living anionic ring-opening polymerisation (ROP) of ethylene oxide (EO). Traditionally, the dispersity of industrially obtained PEG reached up to 1.3, though living polymerisation can narrow the dispersity (Đ) down to 1.01-1.02. While this value seems relatively small, it hides a surprisingly broad distribution. For instance, a sample of PEG 2000 with a Đ of 1.02 still contains over 25 different PEG chains. As molecular weight increases, the complexity grows; with PEG 5000, up to 50 different PEG chains are present in a single sample.
Approved PEGylated drugs predominantly rely on polydisperse PEG,2 such as the PEG 2000 in COVID-19 vaccines and chemotherapy formulations, or the PEG 5000 utilised for protein PEGylation. While their dispersity remains within pharmacopeial requirements for excipients, the heterogeneity of the material creates significant hurdles for precise characterisation and reproducibility. Usually, polydisperse PEG is first modified with reactive groups like amines or thiols.3,4 While the PEG derivative is then purified by liquid-liquid extraction or chromatography, these processes rarely remove the inherent polydispersity or formed PEG impurities. In the final step, when PEG is covalently attached to a biomolecule or combined with a lipid to form nanoparticles, the inherent heterogeneity is further amplified. This leads to the presence of undesired side products and more complex chemical and structural composition of the therapeutic.
The advantages of PEG monodispersity
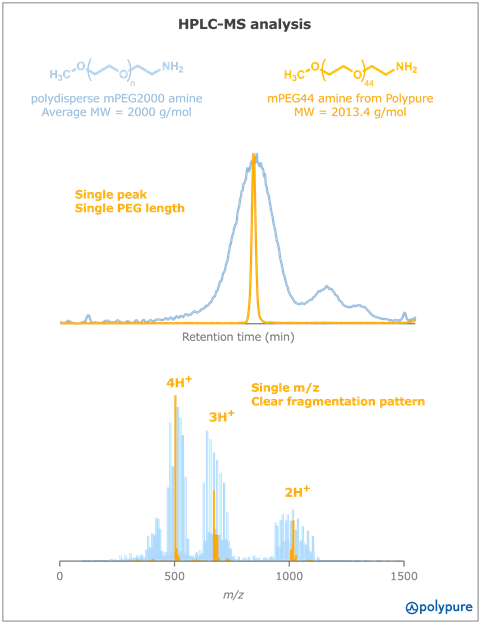
One prominent example of a modified polydisperse PEG is methoxy-PEG 2000 amine (mPEG 2000 amine), widely used for the PEGylation of nanoparticles.5s During the initial ROP of EO, impurities such as dimethoxy PEG or unprotected PEG diol can form. Subsequent derivatisation steps to create activated intermediates, like mPEG tosylate or mPEG bromide, lead to even more contaminants, such as amine dimers or hydrolysed mPEG. Both impurities have been identified via HPLC-MS in a commercial sample of polydisperse mPEG 2000 amine (Figure 1). In contrast, the monodisperse product of Polypure, i.e., mPEG44 amine Polypure is characterised by a single value of molecular weight (MW = 2013.4 g/mol) and high purity (>95%).
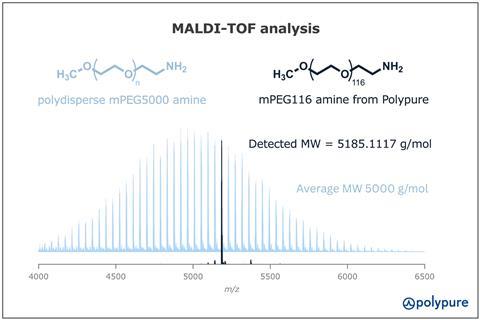
The situation is identical for polydisperse PEG with higher molecular weights, like mPEG 5000 amine. Our MALDI-TOF analysis (Figure 2) reveals an even greater number of chain lengths in polydisperse samples compared to mPEG 2000 amine. With a monodisperse analogue from Polypure, mPEG116 amine the complexity is reduced to a single molecular weight species with MW = 5185.1 g/mol.
The practical limitation of polydispersity was neatly demonstrated by Burggraef et al,6 who compared polydisperse and monodisperse PEG 5000 derivatives for the PEGylation of albumin. The polydispersity of the PEG transferred directly to the final conjugate, preventing the possibility of identifying specific sites of the protein which were PEGylated. The use of monodisperse PEG allowed for the precise identification of these residues. Ultimately, the contrast between the polydisperse and monodisperse PEG highlights how switching from a statistical mixture to a single-molecule species removes the analytical noise and guesswork in the characterisation of bioconjugates.
Enabling precise and reproducible PEGylation
Single-length, high-purity PEG oligomers provide an optimal starting point for further derivatisation and biomolecule attachment. The advantage of using monodisperse PEG has been proven in both commercial products and academic research. Researchers value defined chemical composition in single-molecule imaging and modelling because known chain lengths simplify complex simulations. The primary example are heterobifunctional PEG esters, like Mal-PEG27-NHS ester, perfect as a cantilever in AFM,7 as well as biotin-PEG-thiol derivatives for monolayer formation on gold surfaces.8 In high-volume manufacturing of medicines, like antibody drug conjugates,9 monodisperse PEG linkers, eg azido-PEG7-diglycolic acid, provide high reproducibility, precise control over impurity profiles, and streamlined purification of the final formulation. There is little doubt that medicines based on PEGylated proteins or nanoparticles would benefit from the improved yields and simplified analysis provided by monodisperse PEG. With monodisperse PEG 2000 and PEG 5000 newly available commercially, more consistent PEGylated protein and nanoparticle formulations are well under way.
Find the right PEG derivative for your project
Polypure has the expertise to provide customised products and services for clients seeking specialised solutions in multi-kilo quantities.
References
- J Herzberger et al, Chem Rev, 2016, DOI: 10.1021/acs.chemrev.5b00441
- Y Gao et al, Bioeng Transl Med, 2024, DOI: 10.1002/btm2.10600
- S A Ahmed and M Tanaka, J Org Chem, 2006, DOI: 10.1021/jo0617464
- J Loccufier et al. Makromol Chem, Rapid Commun, 1991, DOI: 10.1002/marc.1991.030120303
- J L Pasek-Allen et al, ACS Omega, 2023, DOI: 10.1021/acsomega.2c07669
- M J Burggraef et al, Nat Commun, 2024, DOI: 10.1038/s41467-024-54076-6
- J Chan et al, Cell Rep Med, 2022, DOI: 10.1016/j.xcrm.2022.100774
- J Rho et al,Biosens Bioelectron, 2018, DOI: 10.1016/j.bios.2017.11.027
- N Joubert et al, Pharmaceuticals (Basel), 2020, DOI: 10.3390/ph13090245














No comments yet