Andy Extance reports exclusively as staff from the €1.5 billion European X-ray Free Electron Laser learn from its first experiments
Leaving his experiment in the underground bunker on the outskirts of Hamburg, Germany, housing one end of the European X-ray Free Electron Laser (XFEL), Christian Bressler makes a snap decision. ‘Come, I’ll show you our setup quickly, before we close the hutch,’ the leading scientist for the femtosecond x-ray experiments (FXE) instrument says excitedly. ‘30 seconds,’ calls one of his teammates.
Bressler points to a short, needle-thin vertical jet of maroon-red liquid open to the air. It is a solution of an iron complex that is part of one of the XFEL’s first two experimental runs. The jet is surrounded by a long line of stainless steel vacuum chambers containing items such as lenses, heavy-duty slits and detectors. Bressler’s team is about to fire an x-ray beam, which will ultimately be a billion times more brilliant than the best conventional sources, at the liquid. While he explains, an automated female voice starts to warn anyone still in the shielded hutch to leave, to avoid being trapped inside and exposed to radiation.

The XFEL’s awesome power promises entirely new capabilities to explore chemistry. Bressler, for example, hopes to simultaneously track atomic and electronic behaviour of substances as they react, seeing events lasting just 15 femtoseconds, or 15 quadrillionths-of-a-second. Other possibilities include determining structures from crystals normally too small for x-ray diffraction, and recording images and movies of biological interactions between substances such as enzymes and potential drugs.
The very first experimental runs started on 14 September 2017 – shadowed exclusively by Chemistry World. Just two of the six European XFEL instruments currently planned were in operation. Their scientists were hard at work balancing the race to publish exciting results with learning how to run an entirely new facility.
Safely back outside, Bressler explains that they are starting by using FXE on a chemical system they’ve studied before. The maroon-red complex forms an unusual iron(V) oxidation state. Now the European XFEL will provide deeper insights about exactly how that happens, to help chemists exploit it. And although a one metre-thick concrete wall separates part of the hardware enabling that from the FXE hutch, it’s covered in a mural showing what’s beyond.
Elephantine electrons
The process of generating the x-rays that the FXE scientists are about to harness begins 3.4km away at the German synchrotron, DESY. The journey starts at the longest superconducting linear accelerator in the world, filled with magnets that direct its electron beam. These crucial magnets were produced in and supplied by Russia as part of a contribution amounting to a quarter of the European XFEL’s €1.5 billion (£1.33 billion) overall cost. Half of the financing for building the facility and provision of specialised equipment was covered by Germany, and the rest is shared by 10 European countries including the UK.
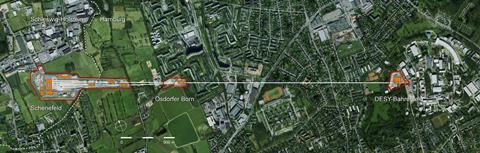
The accelerator will eventually boost around 167 trillion electrons per second up to 17.5GeV. Together, that second’s worth of electrons would therefore possess kinetic energy equivalent an adult elephant running full speed. A further 2.1km down the line they can be switched between two tunnels – although only one is currently operational.
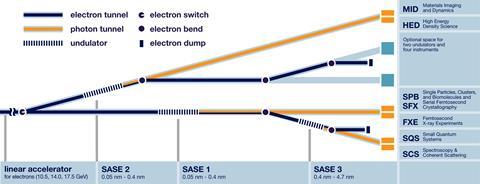
In the final stretch, the electrons pass over a 212m-long ‘undulator’, creating a laser-like coherent beam of in-phase x-rays. Again currently only one undulator, known as SASE1, is running. SASE2, spanning the same wavelengths, and SASE3, offering a different range, will join it by the end of 2018. During the day shift, from 8am to 8pm, SASE1 feeds FXE. During the night shift from 8pm to 8am, it feeds the neighbouring instrument (SFB/SFX), which conducts single particle, cluster and biomolecule experiments, and serial femtosecond crystallography.
10 femtoseconds is fast enough that you effectively freeze atomic motion
Richard Bean, European XFEL
The undulators exploit the fact that passing electrons over a magnet generates radiation, comments SFB/SFX scientist Richard Bean. Ring-shaped synchrotrons like DESY and the UK’s Diamond facility produce x-rays at their beam-bending magnets and at short undulators. But the European XFEL’s undulators align many magnets side-by-side, enabling the self-amplified spontaneous emission (SASE) that gives them their names.
As bunches of electrons go over the undulator they move side to side and radiate x-rays, Bean explains. ‘Eventually, the x-rays align the electrons so that they move together,’ he says. ‘When this happens you get a huge increase in the x-ray emission.’ The resulting x-ray laser beam is also what’s needed to cause the interference effects seen in crystallography. Bean points to a postcard showing dots arranged like a plus-sign, the first diffraction pattern the European XFEL generated. ‘That’s coherent light falling across an aperture,’ he says.
A fine point
Lining up such a pattern isn’t easy because, although the facility that produced it is kilometres long, the powerful x-ray beam focuses down to around two microns in diameter. Both FXE and SFB/SFX are now aligning this with liquid jets with potentially similar dimensions. Bean explains that the SFB/SFX team successfully collected diffraction data from a lithium titanate powder calibration standard on the previous night, day one of their first experimental run. Yet they couldn’t line up their water jet, and are now preparing to try again. The automated setup’s ‘couple of thousand motors and sensors’ will then let the team safely attempt this from outside the hutch, Bean adds.
An hour before SFB/SFX is due to begin day two of its beamtime, Bean and Adrian Mancuso, SFB/SFX’s leading scientist, address around 50 of their colleagues. They’re joined by Anton Barty from the Center for Free-Electron Laser Science (CFEL) based at DESY. Barty devised the first scheduled SFB/SFX ‘community proposal’ allowing the researchers in the room to study biologically significant proteins. And from their questions, it’s clear that the ‘sample guys’ preparing the protein crystals are impatient.

They’re hungry to harness the x-ray bursts that the XFEL accelerator’s narrow electron bunches will produce in 10 femtosecond pulses, Bean explains to Chemistry World. ‘10 femtoseconds is fast enough that you effectively freeze atomic motion,’ he adds. ‘It’s like a strobe.’ In its first experiments, the European XFEL is producing 300 pulses per second, more than twice the Linac Coherent Light Source (LCLS) XFEL in the US. In about a year that could reach 6000 pulses per second, as it works towards 27,000 pulses per second. The gradual ramp-up reflects the necessary precision. ‘If you push this much power in, if the hundreds of pieces that have to be exactly in the beam are not perfectly aligned then you can start doing a lot of damage,’ Bean underlines.
And the need for caution becomes clear when Mancuso reveals that the XFEL’s beam is currently down. One of the tube-shaped superconducting modules has ‘gone pop’.
Diffraction before destruction
The beam went down at the end of the FXE run that had been about to start during Bressler’s tour. In its iron(V) experiment, Bressler’s team is hoping to take advantage of the large number of x-rays the XFEL produces to do both diffraction and x-ray emission spectroscopy.1 This will enable them ‘not only to learn about what the atoms do but why’, Bressler says.
Similarly, the XFEL’s many intense pulses are crucial to the scattershot-seeming approach in which the SFB/SFX instrument fires them at a jet of protein crystals in solution.2 ‘Sometimes you hit the crystals and sometimes you don’t,’ Bean says. ‘But if you hit enough you get several hundred thousand patterns with crystal diffraction in, and [from that you get] the electron density of the molecule.’
Mancuso adds that the pulses are organised into 10 bunches of 30 per second, in which the pulses are separated by around a millionth of a second. The bunch structure creates a new dimension for pump–probe experiments, which use x-ray crystallography to probe chemical reactions started by a pump laser. ‘In a synchrotron you need to integrate many times over different time steps,’ Mancuso explains. This captures each frame in the process separately by gradually increasing the gap between pump and probe. But because the European XFEL’s ‘got a ton of x-rays’ in closely spaced bunches, he adds, it can record several frames at once.
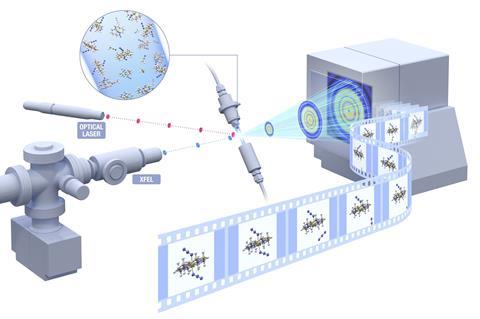
In fact, the x-ray beam is so intense that it destroys every crystal it hits. But because the crystals blow up at the speed of sound, and the x-rays travel at the speed of light, each crystal still yields abundant data before it is blown apart. Yet this ‘diffraction before destruction’ still comes with an aftermath, notes Barty. ‘We know the jets explode, but then we’ve got to bring along the next crystal,’ he says. ‘How do we bring in sample fast enough to get 20 times more structure data in a given amount of time, or 20 time points?’
‘You think the worst’
Barty’s SFB/SFX community proposal therefore aims to help solve this, and in doing so allow various groups to do relatively simple studies on proteins. ‘We know from screening that [the protein crystals] diffract well, but they form very small crystals,’ Barty explains. ‘At LCLS we shot crystals grown in cells, which cannot grow larger than the cell or it dies.3 These you could run at the XFEL quite nicely. Instead of taking 6–12 hours to get a structure, you can start turning them over in minutes ultimately.’
That speed also brings computational pressure. ‘At LCLS you can get half a million diffraction patterns an hour, here we can theoretically go up to measuring 30 million,’ Barty says. ‘We are set to produce tens of terabytes of data per hour. Then you need to be able to crunch through that.’
The community proposal includes the immune system protein myelin differentiation primary response 88, or MYDA88, being studied in Connie Darmanin’s lab at La Trobe University in Melbourne, Australia. MYDA88’s structure has been determined by x-ray diffraction at a synchrotron before, but with difficulty, because it forms long, thin crystals. Their size has thwarted her PhD student Susannah Holmes’s efforts to do more sophisticated studies at synchrotrons that might help in designing drugs for diseases that involve inflammation.
The XFEL’s ability to study small crystals is therefore ideal – if they can actually get the SFB/SFX instrument running well enough to study them. ‘Coming into an experiment where we know it’s commissioning, you think the worst,’ Darmanin admits. ‘It’s always the last few hours in which we get the data.’
Petra Fromme’s Arizona State University team is studying the photosystem plant proteins that use sunlight to turn CO2 into the molecules we eat and the oxygen we breathe.4 ‘Photosystem I consists of more than 150,000 atoms,’ Fromme stresses. ‘There are 288 chlorophylls, this is why it is green in nature. It’s a huge molecular machine and we make little green crystals which we then hope that we will deliver to the beam.’
If we start to see some diffraction data from anything that’s a champagne moment
Adrian Mancuso, European XFEL
Fromme’s team has an SFB/SFX beamtime slot to itself in November 2017, where the scientists hope to kick photosystem I into action with a laser and make a ‘little mini-movie’. But isolating it from algae is an arduous process, taking several months and producing very variable crystals. The photosystem I scientists therefore tagged along on the first experiment in the hope of seeing which size crystals might work best.
Virgin mode
Bean convenes the day three SFB/SFX meeting with the bash of a gong. On day two, expanding the beam to 150 micrometres actually got x-rays to hit their jet’s nozzle, he explains, but the team still couldn’t record water spraying. So tonight’s goal is to see just how the intense x-rays affect the water and hopefully study an inexpensive, abundant protein. ‘If we start to see some diffraction data from anything that’s a champagne moment,’ says Mancuso.
While Allen Orvile from Diamond isn’t directly involved right now, he’s watching and willing the team on because he is leading SFB/SFX’s third experiment, starting on 28 September. His community proposal aims to build on his prior work5 by letting enzyme crystals mix with substrate molecules they’re known to interact with. This can only work with small crystals that the SFB/SFX instrument is well-placed to study, Orvile explains. Additionally, the closely-spaced pulse bunches should be able to capture substrates moving into and out of the enzyme.
When the meeting gong sounds on day four, the news is promising. The previous night, the SFB/SFX team got the jet to intersect with the x-ray beam. And while the sophisticated detectors needed to collect the enormous volumes of high-frequency diffraction data behaved a little strangely, they did record patterns. Consequently, Mancuso and Bean consider setting criteria to help decide which order to try the group’s proteins in. And with another shift due on day five, it seems possible that Darmanin might even see data from MYDA88 before the final hours of the run.
Encountering Bressler leaving his experiment again on day four, his crumpled appearance reflects the challenges of leading the XFEL’s first ‘class’. On the positive side his team has aligned its maroon liquid jet with both the x-ray beam and a laser to enable pump-probe studies. However, they haven’t yet brought the detector online that they need to study electronic structures through x-ray emission. Bressler admits that he’s ‘not sure if we will be able to do new science’ this time.
However, Bressler expected this, and deliberately chose one of his team’s experiments as the one that would suffer as a consequence. ‘We knew that with the completion of XFEL and all the surroundings so soon before the first user experiment it was unfair to put ourselves at the end of the pile, when we know things will work,’ he says. ‘Right now everything is in a virgin mode.’
Yet his excitement still shines through. He’s confident that his team will soon record the kind of transition processes that chemists have previously considered too transient to see. ‘We are actually about to capture this,’ he enthuses.
Andy Extance is a science writer based in Exeter, UK
References
1 A M March et al, J. Phys. Chem. C, 2017, 121, 2620 (DOI: 10.1021/acs.jpcc.6b12940)
2 S Boutet et al, Science, 2012, 337, 362 (DOI: 10.1126/science.1217737)
3 G van der Schot et al, Sci. Data, 2016, 3, 160058 (DOI: 10.1038/sdata.2016.58)
4 C Kupitz et al, Nature, 2014, 513, 261 (DOI: 10.1038/nature13453)
5 F D Fuller et al, Nat. Methods, 2017, 14, 443 (DOI: 10.1038/nmeth.4195)













No comments yet