A ‘one pot’ process for manufacturing oligonucleotides – short 20–30 base pair DNA or RNA molecules – has been developed. Oligonucleotides are of interest as they can be used to treat a range of medical conditions, but mass producing them is still difficult.
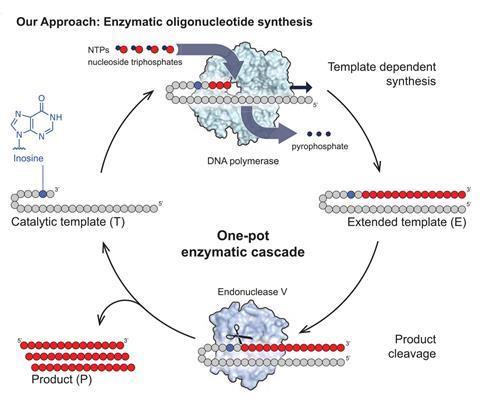
The biocatalytic process to produce oligonucleotides was developed by Sarah Lovelock at the University of Manchester, and colleagues. It can be carried out in an aqueous solution and uses a polymerase enzyme to extend a template strand with unprotected nucleoside triphosphate building blocks and an endonuclease enzyme to release the oligonucleotide, allowing the template to be reused.
The new method is significant, because it could make the manufacture of oligonucleotides cheaper and more scalable than using solid-phase synthesis methods, which have been widely used in industry for more than 30 years.
‘It is definitely a very clever idea,’ says Emily Leproust, an experienced chemist and chief executive and co-founder of Twist Bioscience, a biotech company with a focus on manufacturing synthetic DNA. ‘I think the authors should be commended for doing something that is extremely hard.’
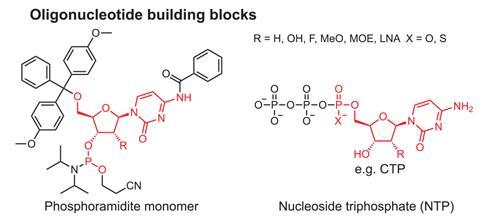
Use of phosphoramidite chemistry to produce oligonucleotides began in 1981, when a paper described nucleoside phosphoramidites. Phosphoramidites have a similar structure to nucleotides, but contain a phosphite not a phosphate group, as well as four other chemical groups that are stable, but can easily be modified. These properties allow different types of DNA and RNA to be artificially synthesised.
While phosphoramidite chemistry was a big advance for industry and is still widely used it has several restrictions. ‘One is scalability,’ Lovelock says. ‘You have these solid supports that you flow reagents through. You can’t just keep making them bigger, because it affects the flow rate through the solid support, which affects the efficiency of the process and then you get a build-up of impurities.’
Leproust explained that while it is technically possible to produce oligos using phosphoramidite chemistry and solid supports on a large scale, it is difficult and expensive. ‘It’s a very fine science to make sure that every piece of the container receives the same amount of chemicals for the same amount of time.’
Phosphoramidite methods also require large amounts of acetonitrile solvent, explained Lovelock, which the new process does not. ‘In order to make one kilo of oligonucleotide, you need about 1000 kilos of acetonitrile. So it’s not very good in terms of green metrics.’
Part of the new generation of advanced molecular therapeutics, oligonucleotides are being used to make therapies such as antisense oligonucleotides and small interfering (si)RNAs. ‘At the moment, most of the approved therapeutics have been for the treatment of rare diseases so they have been produced on very small scale,’ explains Lovelock. But some, such as the recently approved siRNA therapy inclisiran, for treatment of high cholesterol, have a larger patient group and are increasing demand.
Lovelock and colleagues produced several different known therapeutic oligonucleotides, such as pegaptanib, used to treat age-related macular degeneration. Lovelock believes the nature of their technology has the potential to be very scalable. ‘At the moment, we’re at about 2g per litre. Ideally, we want to be above 10g per litre, maybe even 100g per litre.’
The researchers also want to fine tune the protein engineering process to allow a wider range of oligonucleotides to be produced. ‘Oligonucleotides contain lots of chemical modifications. We’ve shown that we have enzymes that can accept various different pharmaceutically relevant modifications, but some are accepted better than others,’ noted Lovelock.
She adds that her team has an ongoing collaboration with Novartis, which owns inclisiran, and AstraZeneca to develop the technology further and test its scalability.
References
ER Moody et al, Science, 2023, 380, 1150 (DOI: 10.1126/science.add5892)














No comments yet