Reaction extends 2010 Nobel prize winning Suzuki coupling and can be used to modify natural products
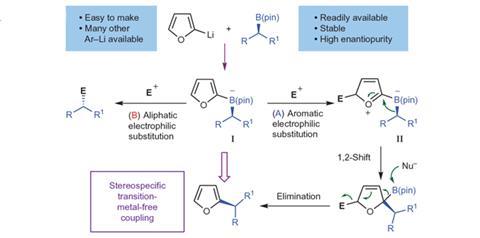
Chemists have devised a new stereospecific coupling reaction for electron-rich aromatics using secondary and tertiary boronic esters that works under mild conditions. The reaction extends the 2010 Nobel winning Suzuki-Miyaura coupling in the synthesis of pharmaceutical skeletons making sophisticated three-dimensional structures more accessible, but without the need for transition metals.
Varinder Aggarwal of the University of Bristol, UK, and colleagues explain that the cross coupling of boronic acids and related derivatives with sp2 electrophiles is one of the most important carbon–carbon bond forming reactions. They are used widely by the pharmaceutical, agrichemical and high-tech chemicals industries. Unfortunately, when it comes to aliphatic boronic esters it is limited to primary compounds and so creates flat products. Secondary and tertiary compounds are not generally open to coupling in this way, but if they were it would add another dimension to such coupling reactions.
The team has now found a workaround that allows them to carry out couplings on secondary and tertiary boronic acids in a stereospecific manner. They used an electron-rich aryl lithium reagent, such as 2-lithiofuran, to convert their boronic esters into intermediate boronate complexes that react with an appropriate electrophile to give a cation. This second intermediate can then undergo 1,2-migration and following rearomatisation and elimination of the boron moiety yields the aryl-coupled product stereospecifically.
Related reactions were attempted more than 40 years ago using achiral symmetric boranes. However, those compounds are not only air sensitive but also lack chirality, which meant that this line of research was abandoned. Aggarwal and colleagues have now, for the first time, exploited readily available, easy to handle boronic acid esters under user-friendly conditions for coupling reactions that could be generalised for many industrial syntheses.
‘We plan to broaden its scope by looking at not just other electron-rich aromatics (boring, but necessary) but also heteroaromatics and even neutral ones too,’ Aggarwal tells Chemistry World. ‘We believe that with the correct substitution and appropriate groups they can all be made to work, thereby providing a new and general method of coupling aromatics to configurationally stable and easily accessible chiral boronic esters.’
‘This is an outstanding process that considerably expands the range of chiral compounds that may be accessed from chiral alkyl boronic esters,’ says James Morken, a specialist in organoboron synthesis and new coupling methods at Boston College, US. ‘The fact that secondary boronic esters participate so selectively is fantastic; that tertiary boronic esters engage, puts this reaction in a league of its own.’ He adds that, ‘Not only does it enable new methods for the assembly of simple chiral compounds and new strategies for the construction of complex targets, as the Aggarwal team aptly demonstrates, it provides a powerful new tool for natural product modification.’
References
A Bonet et al, Nat. Chem., 2014, DOI: 10.1038/nchem.1971












No comments yet