Scientists have created complexes with aromatic three-atom bismuth rings that are stabilised by heavy actinide atoms. These all-metal rings are the heaviest examples of this type of molecule and challenge the idea that aromaticity is limited to carbon-based molecules.
Delocalised organic compounds – such as benzene – define how chemists think about aromaticity. However, there are a number of species that break the classic definitions of this phenomenon. For example, in 2024, scientists synthesised a four-membered bismuth species, making it the first all-metal ring with aromatic bonding character.
Researchers have now added to this family of compounds by creating ‘inverted sandwich’ complexes that feature a three-membered bismuth ring coordinated between two ligands containing uranium or thorium atoms. Such all-metal rings are the heaviest analogue of an aromatic three-membered ring.
Analysis revealed that the bismuth species has a ring current similar to benzene, owing to overlapping σ-orbitals that allow electrons to flow around the ring. This challenges the idea that delocalised π electrons are necessary for aromaticity.
Steve Liddle at the University of Manchester, UK, who led the work, said that such species are ‘a powerful reminder that the deepest principles of chemical bonding apply far beyond carbon’.
References
J Ding et al, Nat. Chem., 2026, DOI: 10.1038/s41557-026-02123-8










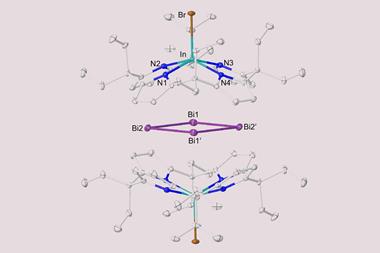


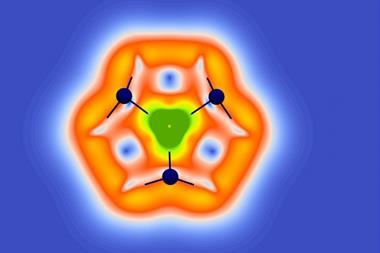










No comments yet