
Methanol has been made in a single step by zapping methane with pulses of electricity. The plasma-based reaction, which also uses water and a copper oxide catalyst at ambient temperatures and pressures, emits no carbon dioxide and could offer a greener, electrified alternative to existing production methods.
Methanol is an important commodity chemical that’s used as a building block for many products, including plastics, paints and adhesives. It is also used as a solvent and as a low emission fuel. Traditionally, making methanol requires two energy intensive steps that first heats methane with high temperature steam, breaking its strong carbon–hydrogen covalent bonds to create syngas, a mixture of carbon monoxide and hydrogen. A catalyst operating under high pressure then turns this syngas into methanol. However, as well as requiring a lot of energy, the reaction itself generates millions of tonnes of carbon dioxide each year.
Now, intrigued by plasma chemistry, Dayne Swearer’s group at Northwestern University, US, has landed on a new electrified route for methanol production. ‘It’s been known for a long time that plasma can do some fascinating chemistry with molecules like methane and water,’ says Swearer. ‘We simply wanted to see if a straightforward approach, like bubbling methane plasma into water, would do anything interesting. To our surprise, that simple experiment produced approximately 40% methanol.’
The team went on to explore the reaction by developing a lab-scale plasma-bubble reactor to control the chemistry. Methane gas was first pumped into a tube in the reactor and zapped with pulses of high voltage electricity. This produced a plasma by freeing some of the electrons from the methane to partially ionise the gas.
The plasma was then forced through a fritted glass diffuser in the tube composed of tiny pores that were lined with a copper oxide catalyst. Passing through the diffuser, the plasma then reacted with deionised water, forming rapidly expanding bubbles and immediately quenching the produced methanol in the water – a process which helped inhibit overoxidation that would make carbon dioxide.
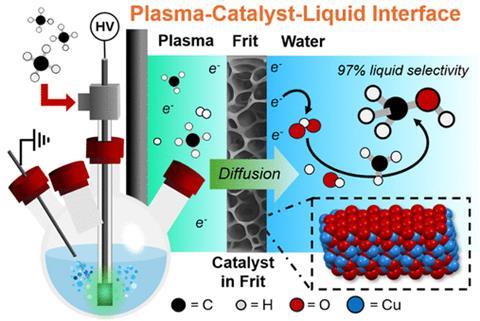
‘In the bubble reactor where the reactions occur in the plasma, on the catalyst, and at the liquid interface, each component has a distinct and important role for making this chemistry work,’ explains lead author James Ho, a PhD candidate in Swearer’s lab. ‘The eureka moment was figuring out how to make all of these complexities work in our favour.’
‘We believe the catalyst helps stabilise specific reactive intermediates, like methyl and hydroxyl radicals, produced exclusively in the plasma zone, enabling higher rates of the desired product, in this case, methanol,’ explains Swearer. ‘We found that controlling how reactive species form and how they move through our reactor enables regulation of reaction outcomes.’
Further experiments showed that diluting methane with argon increased methanol production, reduced unwanted byproducts and achieved a selectivity of almost 97% at the liquid phase of the reaction. Overall, however, the total selectivity achieved for methanol across the entire reaction was around 57%. Results also revealed gas-phase production of hydrogen, as well as hydrocarbons including ethylene and propane, but no carbon dioxide. Just 46.7kW/h of electricity were required make 1kg of methanol.
‘It shows that real breakthroughs may be less about discovering a miraculous new catalyst and more about putting the catalyst in exactly the right place, in this specific case, at the plasma–liquid interface where short-lived reactive species can actually be captured and utilised before they decay,’ comments Andrea Folli, who investigates catalysis for green chemistry at Cardiff University, UK. ‘The selectivity numbers are impressive.’ He says this is more than enough for proof-of-concept. ‘But it is still very far from any meaningful scale that would give important clues on the scalability potential of the technology.’
Ho recognises that it is still early days, with much more optimisation required for scaling up. ‘It is unlikely that this technology completely replaces industrial plants, but it could be deployed for decentralised methanol production where natural gas is being underutilised such as leaky wellheads or sites where methane is just flared,’ he says.
References
J Ho et al, J. Am. Chem. Soc., 2026, 148, 17508 (DOI: 10.1021/jacs.6c04425)












No comments yet