Silicon–carbon bonds have been broken down by a cytochrome P450BM3 enzyme engineered for just that task. The altered enzyme could break the bonds in the mass-produced siloxane, the oligomeric building blocks of silicone, something the researchers say is the first step to making siloxanes biodegradable.
Volatile methylsiloxanes (VMSs) are a type of siloxane that can exist in ring or chain form. They are found in a wide range of consumer products, including textiles, electronics, cosmetics, and medical equipment due to unique properties such as water resistance, flexibility and low chemical reactivity.
In 2018, three cyclic VMSs were identified as ‘substances of very high concern’ under the EU’s Registration, Evaluation, Authorization and Restriction of Chemicals regulations. As they are also highly volatile, VMSs can get into the air, where they can remain for several days.
‘It is important to understand which synthetic compounds can be biodegraded to guide responsible use of the chemicals that enable modern life,’ says Nicholas Sarai, a former PhD student at the California Institute of Technology and first author of study. ‘Our research puts forward the first enzyme that can definitively break the bond between silicon and carbon,’ he adds.
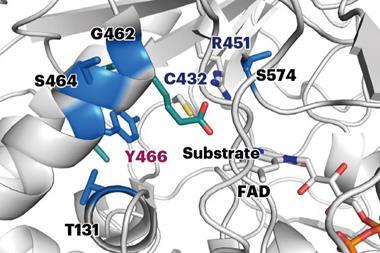
Prior studies suggest that hydroxylation of a C–H in the methyl group adjacent to the silicon in VMS precedes the oxidation – and subsequent breaking – of its silicon–carbon bond. Cytochrome P450 enzymes are known to hydroxylate C–H bonds in alkyl groups. The team hypothesised that it might also be able to hydroxylate the similarly strong C–H bonds in siloxane.
The team picked cytochrome P450BM3 because of its self-sufficiency and high catalytic activity. The researchers then evaluated a number of enzyme variants they created, using a variety of mutagenesis strategies, for their ability to hydroxylate C–H bonds in volatile siloxanes. Following the reaction of a siloxane substrate with these enzyme variants trace amounts of carbinol, a C–H hydroxylation product, and modest amounts of silanol were observed – indicators of silicon–carbon bond cleavage.
The team’s starting enzyme variant showed some silicon–carbon cleavage activity on hexamethyldisiloxane, something not displayed by wild-type P450BM3. Several rounds of directed evolution brought about variants with a better ability to oxidise hexamethyldisiloxane. They then tested variants on a number of different siloxanes, discovering it could break down a number of different types, including cyclic siloxanes.
However, Sarai notes that ‘despite using 5mM siloxane substrate, none of our enzymes are able to generate even 1mM of product.’ But the team says that, underwhelming enzymatic activity notwithstanding, these engineered enzymes show biological activity on VMS is both possible and can be enhanced.
Currently, not all VMS are accessible to these enzymes and the concentration of them in various environments and waste streams is highly variable. ‘The enzymes will need to exhibit higher catalytic activity and different binding kinetics to perform efficiently at these varied concentrations,’ says Sarai.
So although engineered enzymes such as these may eventually be able to clean up siloxane pollution, they must first be engineered to oxidise multiple siloxane methyl groups. ‘Many research questions need to be addressed before these enzymes can be applied,’ says Sarai.
‘Given the ubiquity and magnitude of total siloxane emissions into urban air, their atmospheric lifetimes and potential for bioaccumulation, safely degrading these chemicals is important,’ says Betty Molinier at the University of California, Berkeley, who was not involved in the work. But Molinier points out that the byproducts of these enzymatic reactions, such as formaldehyde, could be harmful from a health and environmental perspective. ‘Although only a small amount of formaldehyde is produced in their experiments, I think it would be useful if future work included more investigation into the quantification of byproducts.’









No comments yet