Intriguing compound has single, double and triple Cr–N bonds from the same chromium atom
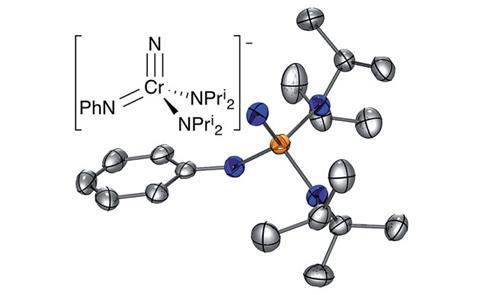
Almost 40 years after Schrock and Clark’s synthesis of a tungsten ‘yl-ene-yne’ complex in 1978, scientists in the US have made the first nitrogen analogue: a chromium nitrido–imido–amido complex.
The team led by Aaron Odom from Michigan State University synthesised the complex with single, double and triple metal–nitrogen bonds to the same chromium atom as part of their research into chromium nitride chemistry. Reacting a nitrido tris(amido) complex with potassium hydride and a cryptand gave amber crystals of the intriguing compound – [K(crypt-2.2.2)][NCr(NPh)(NPri2)2]. They confirmed the bond order using Mayer bond analysis and local natural resonance theory.
Tests on the complex with different electrophiles show electrophilic attack at both the imido and nitrido nitrogen atoms – despite the larger negative charge on the imido nitrogen – so could provide new routes to rare examples of transition metal imido complexes.









No comments yet