Perfume delivery system triggered by water keeps sweet smells around for longer
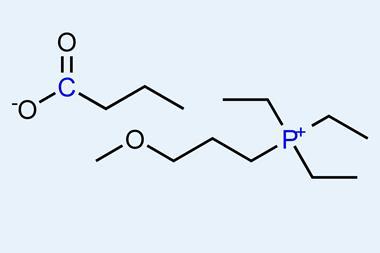
A new perfume delivery system has been developed by chemists in the UK as a way of keeping sweet smells around for longer. This cleverly designed system tags fragrance alcohols – such as 2-phenylethanol, which has a rose-like scent – onto odourless ionic liquids. In the tagged form, the material has no smell. However, when it comes into contact with water, the link is broken and the fragrance is released – along with its sweet scent.
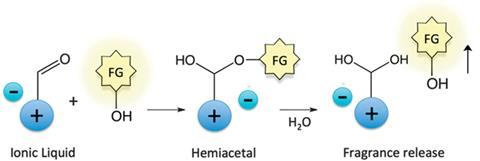
Fragrance alcohols are typically volatile, so their scent can be lost soon after a perfumed product is applied. A lot of research has been dedicated to finding ways to keep scents around for longer.
Critically, the perfume’s rate of release in this study, led by Nimal Gunaratne of the Queen’s University Ionic Liquid Laboratories (QUILL) in Belfast, is dependent on the water concentration. So by controlling the moisture level, the fragrance can be released over several hours – keeping the scent around all day.
Gunaratne highlights that this prolonged release is a key advantage of this system. Although it is early stages, the researchers describe it as ‘an exciting breakthrough’ and are hoping to develop the work further.
One potential application is deodorants, where sweat could provide the necessary moisture. The group have also found that thiols – one component of sweat responsible for its odour – can also trigger the fragrance release. In doing so, the thiol compounds become tagged to the ionic liquid and lose their pungency. This means that in addition to releasing a fresh fragrance, the unpleasant smell of sweat could be simultaneously removed, in a two-pronged attack against body odour.
Christian Quellet, a fragrance delivery expert and consultant with Leugos Sàrl in Switzerland, believes this approach is a promising step in the development of new fragrances. ‘The chemistry involved is innovative, quantitative and atom economical,’ he says.
References
This article is free to access until 29 May 2015. Download it here:
H Q N Gunaratne, P Nockemann and K R Seddon, Chem. Commun., 2015, 51, 4455 (DOI: 10.1039/c5cc00099h)









No comments yet