A new cobalt–tungsten catalyst could significantly boost the efficiency of water splitting without relying on expensive and scarce metals. While other studies focused on tweaking the catalyst composition and improving isolated metrics, the team led by Pelayo García de Arquer from ICFO in Barcelona, Spain, designed a totally new ‘lasagna-like’ layered material that ‘actively involves the ingredients of the reaction’, increasing activity. It’s also an attractive alternative for the generation of green hydrogen, as it showcases a high stability at industrial conditions.

Using renewable electricity to split water yields ‘green’ hydrogen, a clean alternative to ‘grey’ hydrogen obtained by the reforming of fossil fuels. However, state-of-the-art industrial processes rely on scarce ingredients, such as iridium and platinum, key to catalysing the oxygen evolution reaction that takes place at the other electrode, which has been a major bottleneck in improving water-splitting technologies. Chemists are still seeking sustainable solutions to this problem and are looking to more abundant metals, which could cut costs too.
Water’s leading role
‘We found that incorporating water, and water fragments, into the catalyst structure … renders the material more active and more stable,’ explains García de Arquer. To make the material, researchers submerged cobalt tungsten oxide in an alkaline bath, delaminating the structure and incorporating both water molecules and hydroxide ions between the catalyst layers. ‘The lasagna layered structure accommodates water-hydroxide networks … enhancing [its] physicochemical properties,’ he says. It’s like trapping reaction intermediates within the catalyst.
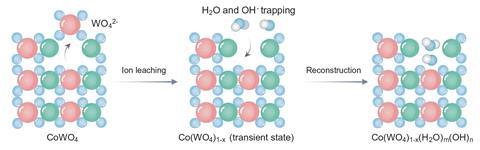
But there is a contradiction at the heart of water splitting: it proceeds more efficiently in acidic conditions but few catalysts can survive this harsh environment with the exception of costly iridium-based ones. This means that industrially most electrolysers operate in basic conditions and take a hit on efficiency. However, in the new catalyst the water fragments shield the cobalt–tungsten metal centres from highly corrosive acidic environments, helping to improve the system’s efficiency.
Until now ‘there haven’t been any promising non-precious metal catalysts for the oxygen evolution reaction’, explains Laurie King, an expert in electrocatalysis at Manchester Metropolitan University, UK. At least, not in scalable conditions, she adds. ‘This paper identifies a transition metal-based catalyst [with] very promising activity and durability in an industrially relevant electrolyser setup.’ In fact, when compared with current catalysts for water electrolysis based mostly on manganese, the cobalt–tungsten lasagna triples the reactions’ current density, linked with productivity and energy efficiency. The catalyst was also stable for over 600 hours when running with a current density and voltage comparable with commercially available alternatives based on iridium oxides. ‘It’s certainly an impressive current density and stability for non-precious metal catalysts,’ says King. ‘This represents a significant leap towards commercial [electrolysers].’
Intriguing design
King also highlights the ‘intriguing catalyst design’ behind the cobalt–tungsten lasagna. To confirm this unusual catalytic structure, García de Arquer and team carried out a comprehensive study combining microscopy, spectroscopy and computational calculations. ‘In particular, in situ Raman spectroscopy was instrumental to assess the presence of trapped water and [confirm] the confined reaction mechanism,’ he says
‘The delamination introduces lattice defects that enable water trapping and hydroxide bridging, which ultimately refine the reaction pathway and the rate-determining step,’ says King. The lasagna actually ‘creates more “active sites”, allowing the mechanism to switch to a less energy-demanding alternative, thanks to the confined water fragments’, says Nuria López from the Institute of Chemical Research of Catalonia, Spain, who coordinated the computational work. ‘We characterised the materials with everything we had at hand [and] simulations were crucial to understand the catalytic process,’ she says.
‘Considering that state-of-the-art materials rely on extremely scarce materials like iridium, discovering non-precious metal catalysts [could] enable the rapid scale up of green hydrogen production,’ says Reshma Rao, an expert in electrochemistry at Imperial College London, UK. ‘Rather than optimising the catalyst’s chemistry alone … the interfacial water structure adds an additional lever to tune activity and stability,’ she notes. On top of pushing performance limits, the lasagna layers are ‘largely responsible for the improved stability in an acidic electrolyte, [because] the trapped water in the structure shields cobalt ions from dissolution’.
However, Rao recognises some downsides too. ‘Although cobalt is more abundant than iridium, it’s also a critical material [with] its own sustainability challenges.’
The authors agree. ‘The issues with critical raw materials are crucial, [we] clearly need to improve sourcing and recycling of the materials in any new technologies,’ explains López. The team will now study several substitutes, including nickel, manganese and other metals, assessing the possibilities of delamination beyond cobalt–tungsten oxide. King adds that this lasagna design could be applied to solve other pressing environmental problems with electrocatalysis, such as turning carbon dioxide into fuels or fixing nitrogen-fixing to make fertiliser production much less energy intensive.
References
R Ram et al, Science, 2024, DOI: 10.1126/science.adk9849
















No comments yet