Touted as ‘soluble sand’, stabilised small silicon oxides present new synthetic worlds
Silicon and oxygen have been harnessed into previously unknown chemistry by US researchers, opening up a new ‘world in a grain of sand’. From soluble disilicon molecules they previously showed helper compounds could stabilise, Gregory Robinson’s team at the University of Georgia have isolated elusive small silicon oxide molecules. ‘Utilizing this carbene-stabilisation strategy, molecular silicon oxide moieties are synthetically approachable,’ says Robinson.
When silicon and oxygen coexist it’s usually in networks and chains, as in quartz and silicones. Because silicon–oxygen double bonds and low-oxidation-state silicon atoms are highly reactive, smaller silicon oxide molecules are generally unstable and rapidly form larger, typically insoluble, groupings.
Since showing that two N-heterocyclic carbenes can stabilise disilicon by donating electron pairs to it in 2008, Robinson’s team has used the approach to isolate several other small inorganic molecules. Yet they harboured a lingering desire to oxidise disilicon. ‘We were recently encouraged by the fact that stabilised diphosphorus was demonstrated in our laboratory to split molecular oxygen, yielding a stable molecule containing diphosphorus tetroxide,’ Robinson explains.
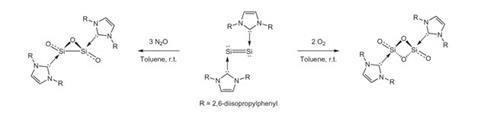
But synthesising small silicon oxides proved to be ‘arduously difficult from a synthetic perspective’. At first glance adding nitrous oxide or oxygen to a stabilised disilicon solution in toluene at room temperature to make Si2O3 and Si2O4 respectively looks straightforward. However, unlike air-stable diphosphorus tetroxide, the silicon oxides immediately decomposed when exposed to extra oxidant.
Despite that sensitivity, the Georgia chemists were able to isolate and retain enough for X-ray crystallography. Together with detailed structural calculations, the crystal structures showed the stabilised Si2O3 and Si2O4 molecules existed with central three-membered and four-membered ring structures respectively.
The work amounts to making sand soluble, enthuses Herbert Roesky from the University of Göttingen, Germany, whose work also exploits carbene stabilisation. ‘This is an amazing result which will have a number of applications in depositing SiO2 on surfaces of various materials,’ he says.
Robinson now hopes to develop new reactions that swap the carbenes for other chemical groups, transferring individual silicon oxides. ‘We also believe that other highly reactive main group oxides may be synthetically approachable using this strategy,’ he adds.
This study will be one of the last to involve the University of Georgia’s pioneering computational chemist Paul von Ragué Schleyer, who died in November 2014. ‘Paul was very excited about this discovery and diligently worked with us right up to his passing,’ Robinson says. ‘His absence leaves a considerable void in us all.’












No comments yet