A computational study has uncovered unusual patterns in the electronegativity and hardness of elements when under the influence of strong magnetic fields.1 The exotic behaviour of chemical systems in this type of environment has long been a topic of interest across chemistry and physics. Now, researchers in Belgium and the UK have extended a method termed conceptual density-functional theory (DFT) to explore them.
Conceptual DFT is a branch of DFT that aims to extract definitions for widely used chemical concepts, like electronegativity and hardness. ‘Gradually conceptual DFT has evolved into a reactivity theory enabling chemists to gain insight into and ultimately to predict the reactivity of molecules under certain conditions,’ explains Paul Geerlings, from the Free University of Brussels. Working with Frank de Proft, Geerlings’ broader research has extended conceptual DFT to include other factors, such as external electric fields and mechanical forces.2,3 ‘It was only natural that we embarked on extending the framework of conceptual DFT to include these,’ says de Proft.
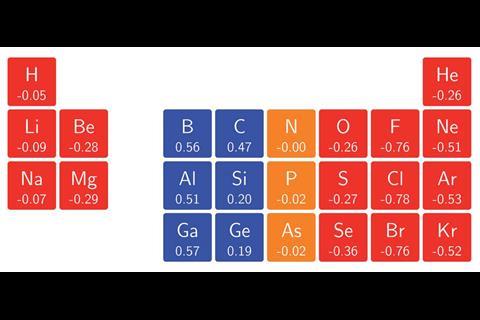
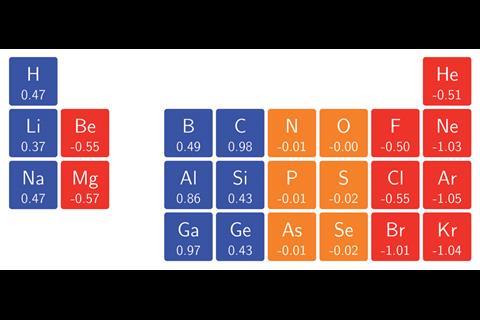
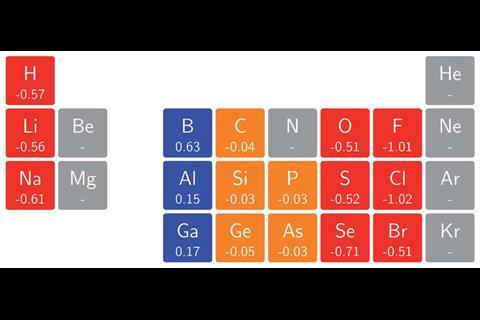
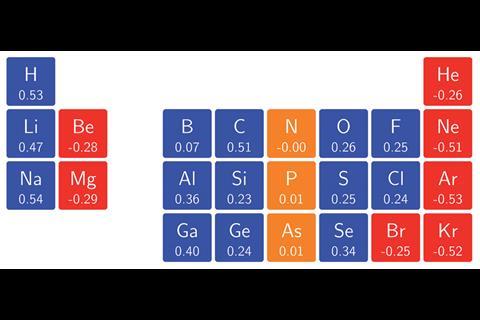
The team surrounding Geerlings and de Proft has now used conceptual DFT to calculate the ionisation energies and electron affinities for main group atoms from hydrogen to krypton in magnetic fields as high as 1B0 (235,000Tesla). In turn, they calculated the electronegativity and hardness, enabling them to construct an alternative periodic table revealing patterns that widely deviate from those expected under no external influence. ‘Perhaps the most surprising observation was how complicated the pattern of ionisation energies and electron affinities, and consequently electronegativity and hardness, with increasing magnetic field strength turned out to be,’ says team member Tom Irons, from the University of Nottingham. This is because the atoms respond to a magnetic field by altering their ground state configuration, with electrons added or removed from different orbitals with varying energies. Despite this, ‘a periodic pattern in the change of electronegativity and hardness with respect to magnetic field strength was still clearly identifiable,’ comments Geerlings.
The researchers hope their findings will enable scientists to predict chemical bonding behaviours under the influence of magnetic fields. ‘For example, the changes in electronegativity of the hydrogen and fluorine atoms as a function of magnetic field suggest that at a critical field strength the polarity of the bond should reverse,’ adds Nottingham team member Andrew Teale. ‘Performing calculations on this molecule supports this prediction — the dipole moment does indeed change direction as the field strength increases.’ Such predictions could even be applied to stellar objects such as white dwarf stars, where typical field strengths reach on the order of 1B0. ‘Certainly, this will be an exciting area for further study,’ says Irons.
Pratim Chattaraj, an expert in DFT from the Indian Institute of Technology Kharagpur, warns that ‘one must be careful as the electronegativity and the hardness are respectively defined as the first and the second derivatives of the energy with respect to the number of electrons at a constant external potential. Once you vary the magnetic field strength, you vary the external potential, defying the definitions.’
References
1 R Francotte et al, Chem. Sci., 2022, 13, 5311 (DOI: 10.1039/d1sc07263c)
2 T Clarys et al, Phys. Chem. Chem. Phys., 2021, 23, 990 (DOI: 10.1039/d0cp05277a)
3 T Bettens et al, Phys. Chem. Chem. Phys., 2019, 21, 7378 (DOI: 10.1039/c8cp07349j)











No comments yet