A new type of permanently porous liquid based on metal–organic cages has been created. The materials are recyclable and can encapsulate both gaseous and non-gaseous guests, which makes them attractive for catalysis and chemical separation.
While permanent porosity is a property that is usually associated with solids, in recent years scientists have been able to engineer persistent cavities into liquids using different approaches. Until now, the applications of these ‘permanently porous liquids’ have focused on gas storage and separation – and most studies have been limited to the binding of small molecules such as carbon dioxide and methane – so porous liquids with holes that can host more diverse guest molecules are in demand.
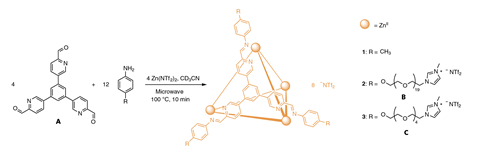
Researchers working with Jonathan Nitschke of the University of Cambridge, UK, have taken an important step in this direction by preparing coordination cages that behave as a liquid in the neat state and can bind many more guests than the porous liquids reported so far. Their study builds upon earlier work by a team led by Stuart James of Queen’s University Belfast and Andy Cooper at the University of Liverpool, UK, who reported the first permanently porous liquid in 2015. ‘That work is fantastic and inspired us greatly,’ Nitschke says. ‘One key difference is that their system has two components: 15-Crown-5 [crown ether], which acts as a solvent, and an organic cage, which provides porosity. Our system is composed of only a single ionic liquid.’
The new material consists of hollow tetrahedral cages with a zinc ion at each corner of the tetrahedron. ‘Three fluidifying oligo(ethylene glycol) chains are grafted onto each corner, and each of these chains is capped with an imidazolium group,’ explains Nitschke. James points out that metal–organic cages are a diverse class of structures, so this approach will enrich the properties of porous liquids. ‘They used a neat trick to keep the long chains out of the pores electrostatically, by making their ends positively charged just like the cage itself,’ he says.
The scientists also investigated the binding of different guests within the empty cavities of their new liquids. ‘Both size and shape selectivity was observed for a range of isomeric alcohols, and the uptake of ozone-depleting chlorofluorocarbons was studied,’ says Becky Greenaway of the University of Liverpool. She believes that the work could open up new avenues for selective and recyclable molecular separations. ‘To the best of my knowledge, this is the first tangible example of a true molecular type-I porous liquid,’ Greenaway says. ‘That is, it maintains permanent porosity in the liquid state and does not require the use of a solvent.’
‘This is exciting work,’ says Cooper. ‘To me, the key advance is the preparation of porous molecular liquids with negligible vapour pressure. This opens up the use of heating or vacuum to release the guests after they are captured. The cages that we reported in our 2015 paper were also non-volatile, but the bulky carrier solvents were not.’ Cooper believes that a possible limitation of the new materials could be their viscosity, which may prohibit some processes. ‘But the move to negligible volatility materials is an important step forward,’ he says. James adds that coordination cages can be quite labile, so the porous liquids might not be that robust. ‘Also, as ever with realistically considering long-term applications, the cost of synthesis also needs to be considered. However, at this early stage in the field, proof-of-principle work such as this is highly valuable.’
The team is now planning to prepare liquids with even larger cavities that can bind more complex guest molecules.
References
L Ma et al, Nat. Chem., 2020, DOI: 10.1038/s41557-020-0419-2









No comments yet