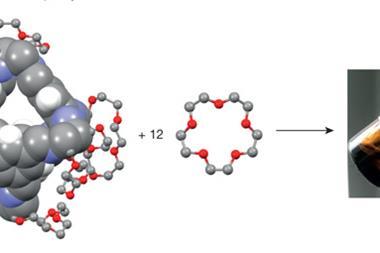
Organic cages have been used as precursors to synthesise higher-order porous structures, adding to their functionality while the ability to solution process them is retained.
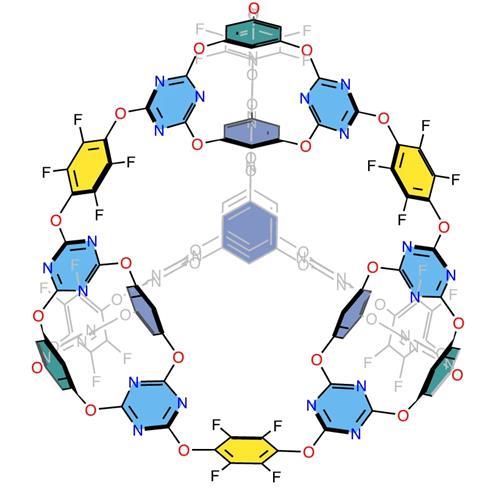
The team from the UK and China used ether-bridged cage molecules as a building block – its chlorine atoms are essential for forming ether bridges – with fluorine-enriched tetrafluorohydroquinone (TFHQ) as the linear bridge. The fluorine atoms offer structural integrity by limiting bond rotation and can improve the solubility of the resulting cage–cage molecules.
Models were constructed using molecular dynamics and density functional theory to predict the reaction products of these blocks. Several topologies and their relative energies were predicted, not considering solvent effects, and the results indicated a strong preference for a [4[2+3]+6] cage product. The team, guided by these simulations, conducted two-step assembly experiments to screen optimum conditions.
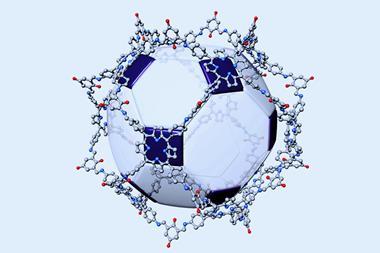
The results showed good agreement between the predicted structure for the [4[2+3]+6] cage molecule and the observed crystal structure – four trigonal cages assembled into a larger tetrahedral cage. The product demonstrated both good sorption capacity and hydrolytic stability – important properties for gas separation and water remediation technologies.
This new ‘cage of cages’ structure could be used as a building block for even more complex structures. This study highlights the use of computational methods to assess the most likely reaction products as well as non-intuitive new materials in supramolecular synthesis.
References
Q Zhu et al, Nat. Synth., 2024, DOI: 10.1038/s44160-024-00531-7












No comments yet