IBM researchers create polymers that breakdown in acidic conditions
Unlike thermoplastics, in which the polymer chains are not chemically bonded to one another, thermosetting polymers are highly cross-linked, so the chains can’t be untangled when the polymer is heated. This crosslinking gives them better physical strength and chemical stability – qualities valued in industries from microelectronics to aeronautics – but it also makes them extremely difficult to recycle, so large quantities are thrown away.
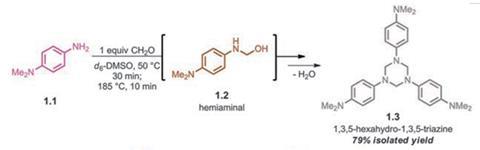
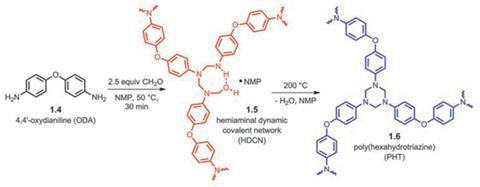
Jeanette García at IBM Almaden Research Center, US, and colleagues designed and synthesised two new classes of thermosetting polymer from difunctional amine monomers that polymerise into a triangular network in a paraformaldehyde condensation reaction. At temperatures around 50ºC, flexible polymers cross-linked by hemiaminal dynamic covalent networks (HDCNs) are produced. At higher temperatures, stronger, more brittle polymers linked by rigid triazine links are formed. One such material, poly(hexahydrotriazine) (PHT) is one of the strongest thermosets known and was made even stronger by incorporating 2–5% carbon nanotubes into it.
Hatsuo Ishida of Case Western Reserve University, US, describes the work as ‘a groundbreaking proof of concept’ although he believes the specific materials demonstrated here would probably be unattractive to engineers. For example, he says, the PHT fails at only 1% strain, which would make it problematic as a matrix material in a fibre-reinforced composite. ‘You need to have the resin fail after the fibre fails, otherwise you will not be able to utilise the fibre strength fully,’ he says. ‘From that point of view, this matrix polymer is far too brittle.’













No comments yet