The polytope formalism – originally developed to systematically describe stereoisomerism – has now been expanded to explore the exact arrangement and connection of atoms within a molecule, aka molecular constitution. The unconstrained nature of the formalism allows it to explore regions of chemical space inaccessible to other methods.
Isomerism and stereoisomerism are central to numerous chemical and biological processes, and understanding how molecules transform into their various isomers could help chemists better tailor reactions and products. But the space of possible molecular configurations is vast, making it challenging to explore fully.
Traditionally, molecular constitution is described using canonical representations, which depict atoms and their bonds, including bond multiplicities. While this approach is useful, it struggles to capture bond formation processes during intermediate states of chemical reactions.
By contrast, an atom connectivity representation without any additional assumptions allows chemists to capture the full spectrum of possible configurations for a given set of atoms, including reaction intermediates and association–dissociation processes. In this representation, each configuration corresponds to a connectivity matrix whose rank equals the number of atoms, with entries of 0 or 1, where the 1 corresponds to a connection between the pair of atoms. Enumerating these matrices yields a rigorous mathematical description of chemical space, although the number of possible configurations grows super-exponentially with the number of atoms.
The polytope formalism provides a way to organise and navigate this immense configuration space. Originally developed to systematically describe stereoisomerism, it treats molecular arrangements as nodes in a graph-like landscape and shows how they relate and interconvert. Now, Peter Canfield and Maxwell Crossley at the University of Sydney in Australia have extended these principles to the much larger problem of mapping every possible connectivity pattern between atoms – not just their spatial arrangements – creating a framework capable of linking molecular structure, reactivity and mechanistic pathways.
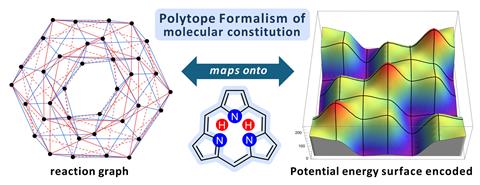
The formalism combines graph theory to establish properties of the configuration space with theoretical calculations to encode the potential energy landscape of the configurations. As Crossley puts it, ‘the formalism deals with the topology of chemical space whilst the calculations add the topographic information.’
To reduce the computational complexity of the problem, Canfield and Crossley relied on modular structure theorem to impose constrains on the permitted transformations, resulting in reaction graphs that can be thoroughly analysed using energetic calculations. They also introduced a taxonomic hierarchy – class, family, genus and species – to organise the outcomes. The result is a comprehensive tool to explore chemical space ranging from general structure to deep mechanistic insights with potential for automated exploration and classification.
Canfield says their work will help bridge the gap between theory, experiment and synthetic approaches. ‘Not only does the formalism comprehensively and rigorously define chemical space at a granular level, but it also gives you a very efficient way of exploring it using simulation approaches.’
‘Designing new molecules can take months to years, and you might not know whether they are going to work in the end,’ says Cranfield. ‘Meanwhile, it is quite quick and cost effective to simulate the properties of an intended target and help guide the synthetic chemist to choose their synthetic targets.’
Christina Moberg, an organic chemist from the KTH University in Sweden, calls this extension of the polytope formalism to molecular constitution ‘a major step forward’. ‘The formalism no doubt has potential also for more general applications to chemical processes, thus providing a more complete unifying framework.’ She notes that the method’s ability to explore chemical space without constraints is one of its strengths, though this openness can also introduce new challenges: ‘The formalism has the advantage of not being limited to solutions a priori considered as “reasonable”, the outcome of the exploration of the entire chemical space may be considered too complex for further analysis. In addition, inherent limitations caused by exceedingly complex problems may appear.’ In Moberg’s view the most significant contribution of the method is it wide appeal since ‘the principles behind the formalism, which are possible to grasp without mathematical formalism, will influence the way a broad range of scientists think.’
References
This article is open access
P J Canfield and M J Crossley, Chem. Sci., 2026, 17, 2102 (DOI: 10.1039/d5sc08813e)









No comments yet