Moses Gomberg, often referred to as the ‘father of radical organic chemistry’, may have unknowingly made a trivalent carbon years before he reported the discovery of these chemical species in 1900.1 Chemists who recreated one of Gomberg’s experiments say that the findings could have strengthened his evidence that carbon could only have three bonds, leading to earlier acceptance by the chemistry community.
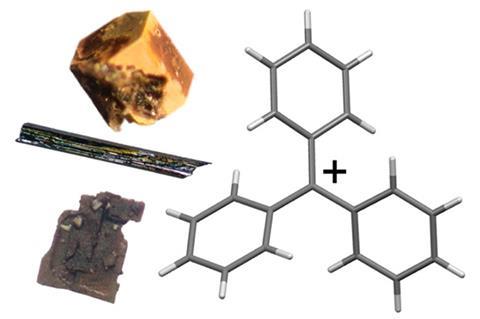
Gomberg was a Russian-born chemist who studied chemistry at the University of Michigan in the US in the late 1800s. He spent the early part of his career in Germany working out how to synthesise tetraphenylmethane, which was ‘a real synthetic challenge’ says Bart Kahr, a crystallographer at New York University in the US. After returning to Michigan, Gomberg attempted to make the next member in the series – hexaphenylethane. ‘His inability to make that ostensibly simple and stable molecule is what led him on the trail to a reactive free radical,’ says Kahr.

One of Gomberg’s later attempts to synthesise hexaphenylethane involved reacting triphenylmethyl halides with silver in benzene, producing a white powder that readily reacted with oxygen and halogens. These properties were unexpected, with Gomberg concluding in his findings that ‘we have to deal here with a free radical – triphenylmethyl’.2 He proposed that the hypovalent carbon atom was in equilibrium with its dimer, hexaphenylethane.
‘In those days, all we had in chemistry were valency rules – nitrogen makes three bonds, carbon makes four bonds, and so on,’ explains Kahr. ‘And so he was proposing to knock out the foundation of structural chemistry by violating valency rules.’ Gomberg’s idea met fierce resistance from other researchers, and it wasn’t until the 1930s that free radicals became accepted by the organic chemistry community.
Revisiting Gomberg’s experiments
Kahr and his team stumbled on some of Gomberg’s earlier work while trying to synthesise herapathite, a dichroic material that polarises light. Polaroid used herapathite in its first photographic sheets.
‘I netted this Gomberg paper from 1898 in which he was trying to make a simpler analogue of herapathite. And the chemistry was so easy – you just mix triphenylbromomethane, iodine and benzene [together],’ explains Kahr.
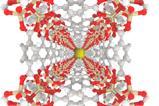
Gomberg originally thought that the black crystals were a molecular complex of triphenylbromomethane and iodine. However, when the US team repeated Gomberg’s experiment, analysis by x-ray crystallography revealed that there were three different crystal structures.
Each of the three crystals contained the triphenylmethyl cation, but alongside varying ratios of iodine and bromine. As these carbocations are less reactive than the radical equivalent, and subsequently exist for much longer, it may have been easier to use such species to convince the chemistry community of the existence of hypovalent carbon, explains Kahr.
‘X-rays were discovered in 1895 and the electron in 1897, so Gomberg didn’t have x-ray crystallography [as a tool],’ says Kahr. ‘[Gomberg] characterised his elemental iodine in a variety of ways but he didn’t distinguish crystals that were of different shape … he treated all of these black solids as equivalent.’
Peter Schreiner, an organic chemist at Justus Liebig University in Germany, says that the discovery offers hope for chemists everywhere. ‘It is reassuring to observe that even prominent figures such as Moses Gomberg, in a single instance, failed to fully comprehend their experimental findings,’ he says.
References
1. C Grainger et al, J. Am. Chem. Soc, 2026, DOI: 10.1021/jacs.5c21781
2. M Gomberg, J. Am. Chem. Soc., 1900, 20, 757 (DOI: 10.1021/ja02049a006)













No comments yet