Several stable radical chains with multiple nitrogen atoms have been synthesised, with some persisting at room temperature for several weeks. The researchers behind the work say that this stability may allow such compounds to act as a storable source of nitrenes, a typically hard-to-contain reagent that can be used to synthesise nitrogen-containing drugs and natural products, for example through C–H activation.
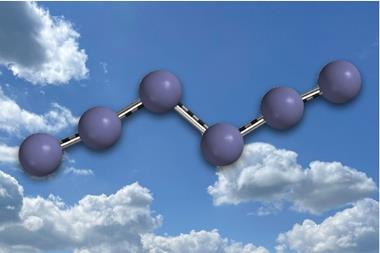
Intense solar radiation in the Earth’s ionosphere can break the strong dinitrogen triple bond, allowing nitrogen molecules to fuse together to form chains. However, under ambient conditions, such species are highly reactive – rapidly breaking down into dinitrogen gas – and are only stable under extremely high pressures of at least 10GPa.
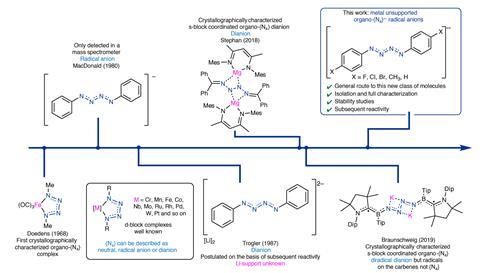
Scientists at the University of Manchester and Oxford have now synthesised several linear nitrogen radical chains with four nitrogen atoms at room temperature, without the need for metal cations to stabilise the structure. To do this, the team reduced para-substituted phenyl azides with potassium graphite, where substituents included fluorine, chlorine, bromine, methyl and hydrogen.
The brominated compound remained stable for several weeks under sealed conditions, allowing the team to analyse the delocalised electronic structure of the nitrogen chain experimentally using spectroscopic and fluorescence-based techniques. This was previously difficult to do, as these molecules rapidly decompose or react with other compounds.
The team were also able to activate the C–H bond of 4-iodobenzaldehyde with the brominated compound, creating a molecule with an amide bond. Due to the increased stability of these nitrogen chains, the team says that these compounds could be a storable source of nitrene radical anions.
References
R Lister-Roberts et al, Nat. Chem., 2026, DOI: 10.1038/s41557-025-02040-2














No comments yet