Artificial cells have previously lacked the complexity that is distinct to natural life but scientists are edging closer
Researchers in the UK have engineered self-reproducing lipid protocells with advanced and dynamic functions. The work gives weight to the importance of lipids in life’s origins but also hints at new functionalities for supramolecular chemistry.
Many research teams have developed artificial systems with properties like self-reproduction, compartmentalisation and adaption to external signals in isolation. Integrating several of these properties into the same system, however, is more difficult.
Now, Stephen Fletcher and Elias Post at the University of Oxford have made a self-reproducing lipid vesicle model that features an in situ organocatalyst, created under specific environmental conditions.
Phase out
Vesicles’ ability to form barriers enables several cellular functions. In the system devised by Fletcher and Post, phase-separated hydrophobic and hydrophilic starting materials, along with an additional copper catalyst, react together slowly to form an amphiphilic phospholipid. After reaching a critical concentration, the phospholipids self-assemble into bilayers, which then accelerate their own formation in an autocatalytic cycle.
In the natural world, systems require continuous inputs of energy and maintain an out-of-equilibrium state. This is indispensable to life but very tricky to control in artificial systems. Fletcher and Post enabled vesicles to mimic the out-of-equilibrium conditions of life through dissipative self-assembly.
To do this, they introduced a hydrolysis step that destroyed the phospholipid. Instead of the thermodynamic product forming, it is destroyed at the same rate as it is created, holding the system out of equilibrium. However, to stop a build-up of undesirable waste, and to maintain the level of building blocks needed for the reaction to run continuously, a syringe pump added starting material and removed waste from the reaction mixture.
‘Maintaining autocatalysis in out-of-equilibrium systems will likely open up additional avenues for exploring how environmental selection pressures can alter the composition of formed protocells,’ says Neal Devaraj from the University of California, San Diego in the US, whose research involves synthesising artificial membranes.
After establishing the protocellular system, Fletcher and Post looked at what would happen if they put shorter lipid tails into the starting mixture and were surprised to find that micelles out-compete vesicles in neutral conditions. However, in basic conditions vesicles are the dominant supramolecular structure formed.
Fletcher says the interesting selection behaviours are because the short-tailed micelles take up the copper catalyst into the aqueous phase. This causes phase separation of the catalyst away from the long-tailed starting material in the organic phase, inhibiting the vesicles from forming.
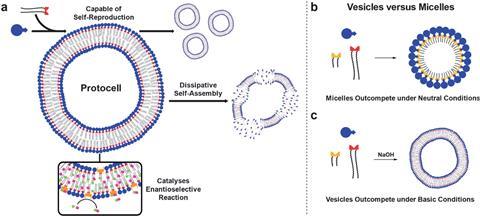
The study also demonstrates that it is possible to use vesicles to catalyse another reaction in situ, effectively creating a type of nanoreactor. The cascade of events shows that these simple lipid networks could be used as a type of molecular machine with advanced functions.
‘Overall, the work shows that under dissipative non-equilibrium conditions there are many interesting changes in self-assembly as well as in catalysis that are different from what one would expect,’ comments Thomas Hermans, an expert in dissipative nonequilibrium self-assembly at the University of Strasbourg in France. ‘Obtaining more life-like systems is an exciting new direction in supramolecular chemistry.’
Life’s containers
Protective bilayers control reactions by concentrating materials and is theorised by the Lipid World model as being a key contributor to the emergence of life.
The study ‘provides strong support to an emerging alternative view of life’s origin, suggesting a central role for simple self-reproducing lipid assemblies, in contradistinction to overly-complex self-copying biopolymers,’ comments Doron Lancet, a human geneticist from the Weizmann Institute of Science in Israel and father to the Lipid World model. ‘I am delighted to see experimental evidence providing strong support to our model, which formally shows how, without genetic material, micellar aggregates can portray non-equilibrium dissipative dynamics, leading to life-defining self-reproduction.’
References
This article is open access
E A J Post and S P Fletcher, Chem. Sci., 2020, DOI: 10.1039/d0sc02768e











No comments yet