‘Atoms’, a student of the English chemist Henry Enfield Roscoe asserted in the late 19th century, ‘are round bits of wood invented by Mr Dalton.’ He was referring to the models created for John Dalton to illustrate the atomic theory described in his 1808 book A New System of Chemical Philosophy. Dalton made concrete (or rather, wooden) the centuries-old idea that all matter is composed of fundamental and indivisible particles, arguing that these invisibly small building blocks combine in fixed and generally simple proportions to make molecules.
Ernest Rutherford’s solar-system model of the atom, proposed a century after Dalton’s book, rendered the atom far less solid than those wooden spheres. Instead, it seemed they were mostly empty space, bounded by electrons that orbited the central dense nucleus. All the same, the electron orbits seemed to impose a sharp boundary where the atom began and ended. But quantum theory soon showed that electrons don’t really have orbits at all, but instead comprise clouds of probability – orbitals – that decay smoothly (albeit in complicated geometric forms) with increasing distance from the nucleus. The quantum atom has no real edge.
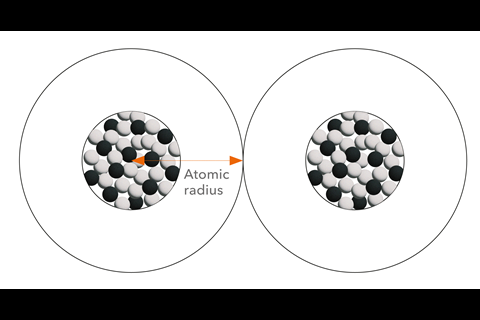
And yet the notion of atoms as spherical objects with particular radii remains important in chemistry. Atomic and ionic radii have long been used, for example, to understand how they pack together in the solid state. Even if we know that the notion of an atom stopping at some distance from the nucleus is artificial, it seems to be a useful approximation. ‘We chemists tend to idealise atoms as hard spheres that may exist independently or connected by sticks with other atoms to form molecules, chains or networks,’ says inorganic chemist Santiago Álvarez of the University of Barcelona in Spain.
But in reality ‘there is no abrupt boundary telling us precisely where an atom ends,’ Álvarez adds. So ‘where exactly should one draw a boundary and say: this cutoff value defines the surface of the atom or molecule?’ asks biophysical chemist Lars Schäfer of Ruhr University of Bochum in Germany. Can any such choice be universal, or must it depend on the context in which we evoke it? And which is the ultimate arbiter: (quantum) theory or experiment?
There’s still no consensus. ‘We have some models and theories [of atomic size] but none have been really amenable to experimental verification,’ says Amin Alibakhshi at the Technical University of Dortmund in Germany. ‘That’s why we have many different definitions, like van der Waals radii, covalent radii, and so on.’ But is each of these definitions in the end a rather arbitrary attempt to carve up the smooth and continuous electron density so that it seems to have hard edges? Or might there be, after all, some deeper and more objective meaning to the size of an atom?
Russian doll atoms
The physical and chemical properties related to atomic radii are too numerous to list; they include electronegativity, hardness, ionisation potentials, melting points, porosity, electrical conductivity, and the characteristics of layered materials. It’s not surprising, then, that the size of atoms has been the subject of a huge amount of theory and experiment, going back as far as Lothar Meyer’s studies in 1870 of how atomic weights of elements relate to their properties.
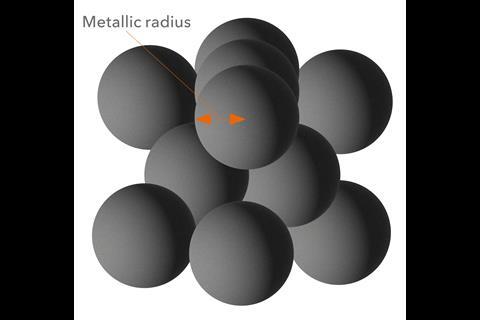
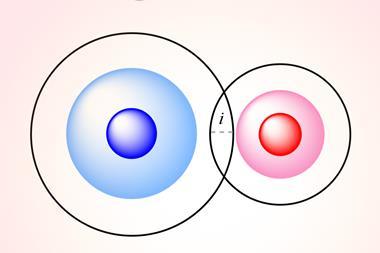
A little chemical consideration suggests that there is unlikely to be literally a one-size-fits-all for any given atom. For example, it seems unlikely that the effective size of an atom will remain the same regardless of which other atoms it is bonded to – in a polar and nonpolar molecule, say. Álvarez says he thinks of atoms as being comprised of a set of three concentric spheres: ‘All atoms have three regions from the centre out, with high, intermediate and low electron densities, even if there is no clear-cut separation between those regions.’ These, he says, can be labelled, respectively, the core, the valence region and the van der Waals territory, each with a specific radius. ‘So we have three atomic radii’ for each atom, he says (the exceptions being hydrogen and helium, which have no core).
This anatomy of atoms is useful, Álvarez says, because each region can be considered to have a particular role in atomic interactions. The core does nothing much at all: the electrons there ‘are inert as far as chemical bonding is concerned’. The valence region, meanwhile, is where chemical bonds are formed, whether those are covalent, ionic, a mixture of both, donor–acceptor, or whatever. The van der Waals radii correspond to the distance at which atoms can interact with one another through the van der Waals interaction – which again decays smoothly (as an inverse power law in the distance from the nucleus) but which can in effect be considered negligible beyond a particular radius.
One atom, many sizes
The atomic radius concept only makes sense in context: how it is defined and measured depends on an atom’s chemical environment. Different definitions are useful for different purposes, and even the same definition might have different values when a definition has changed or as measurements improve.
One atom, many sizes
The atomic radius concept only makes sense in context: how it is defined and measured depends on an atom’s chemical environment. Different definitions are useful for different purposes, and even the same definition might have different values when a definition has changed or as measurements improve.
Atomic radii explanations
This visualisation shows different definitions of the atomic radius of hydrogen.
- H ionic radius — 1.54Å: Meaningful values emerged in the 1950s, with the rise of neutron diffraction.
- Covalent radius — 0.37Å: Calculated by Linus Pauling in the early 1930s by analysing experimentally measured bond lengths.
- Bohr radius — 0.529Å: The average distance between the electron and the nucleus in the ground state of a hydrogen atom. Calculated by Niels Bohr using an early quantum model.
- Van der Waals radius — 1.25Å: Calculated by Arnold Bondi in 1964 by analysing experimentally measured closest non-bonded distances between atoms in molecular crystals and liquids, then assigning each atom a radius equal to half the minimum contact distance where no covalent bonding occurs.
- Empirical atomic radius — 0.25Å: Obtained by John Slater in 1964 by fitting experimental interatomic distances from crystals and molecules, assigning each atom a radius so that the sum of the radii reproduces the observed bond lengths.
- H– Van der Waals — 1.67Å: Determined by Pengfei Li and colleagues by considering the electron density contour at a cutoff of 0.0015 atomic units.
- Van der Waals — 1.20Å: Theoretical value based on atomic polarisability from Jorge Charry and Alexandre Tkatchenko.
- H absolute radius — 1.20Å: The most probable radius of the outermost orbital, obtained via quantum mechanical calculations by Richa Khatiwada and Pengfei Li.
- Van der Waals — 1.20 Å: Value obtained via statistical analysis of non-bonded contacts in crystal structures by Santiago Alvarez.
The atom’s edge
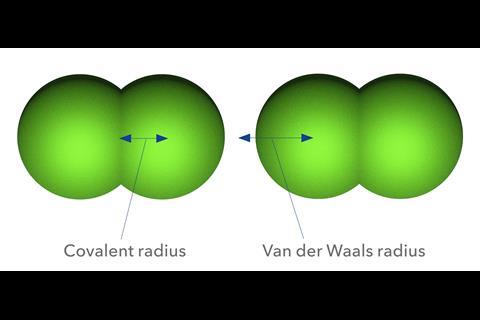
The van der Waals radius is the easiest to conceptualise. To a first approximation, two atoms that don’t engage in bonding can’t approach closer than this. So it’s a measure of an atom’s size in isolation, which we can define by choosing a cut-off value of electron density where we say the atom stops. In 1967 Canadian theoretical chemist Richard Bader and co-workers proposed a cutoff value of 0.002 electrons per bohr-3 (1 bohr = 5.292´10-11 m).1 10 years later another Canadian theoretical chemist, Russell Boyd of Dalhousie University, argued that a better cutoff is just half that value.2 Recent calculations using density-functional theory to deduce the electron distributions for all the elements up to 96 (curium) show that Boyd’s choice of cutoff gives sizes that agree well with those values derived from crystal structures.3
Maybe van der Waals radius is more fundamental than people think
Alexander Tkatchenko, University of Luxembourg
The idea of a van der Waals radius goes all the way back to Dutch scientist Johannes Diderik van der Waals himself, who in his 1873 thesis at the University of Leiden in the Netherlands modified the ideal gas law describing the relations of pressure, temperature and volume of a gas to allow for the fact that the molecules have a finite volume, described by a parameter that basically quantifies the molecular radius. The actual volume available to the gas molecules is therefore smaller than that of the container that holds them, because of this excluded volume of the molecules themselves.
Given that the electron density of atoms and molecules decays gradually with distance from the atomic nuclei, what characterises this excluded volume? In 1964, Arnold Bondi, working at the Shell Development Company in Emeryville, California, US, proposed that van der Waals radii could be assigned to atoms based on the packing densities for the crystal structures extrapolated to absolute zero, where the size wasn’t artificially inflated by thermal motions.4
Bondi’s values were used for many years, and sometimes still are. But he offered values only for 28 of the main-group elements. Donald Truhlar of the University of Minnesota and colleagues filled in the gaps for the remaining 16 main-group elements in 2009, using quantum-chemical calculations to deduce how close to the atom the probe molecules HF and CH4 can approach.5
As that exercise implied, the Bondi radii are basically a measure of when two atoms start to repel one another as they approach in the gas phase. Arguably a more chemically meaningful measure of size is the region within which solvent molecules can’t penetrate in solution – the solvent excluded surface. This is generally bigger than the van der Waals volume, but it’s not clear that there is a unique way to define it for all situations. Alibakhshi, working with Bernd Hartke of the University of Kiel in Germany, has shown that such molecular surfaces can be deduced from a theoretically predicted relationship with the enthalpy of vaporisation of the substance (more specifically, with its temperature dependence) – something that can be measured experimentally.6 They argued that this approach can define a thermodynamically effective molecular surface which, while only slightly different from the van der Waals surface defined by earlier methods, is more accurate for predicting thermodynamic properties.
What do such surfaces mean physically? Alibakhshi and Schäfer have recently shown that it is possible to assign a cutoff value for electron density, calculated using state-of-the-art quantum chemical methods, that gives atomic radii consistently in agreement with the values obtained from Alibakhshi’s thermodynamic method.7 Interestingly, the cutoff value recommended by this approach – about 0.0016 electrons bohr-3 – is about midway between those proposed by Bader and Boyd.
Condensed-matter physicist Alexander Tkatchenko of the University of Luxembourg and his colleague Jorge Charry recently looked at a range of different definitions of the van der Waals radius and found that they show a surprising degree of agreement, even though the definitions themselves look quite different.8 ‘So maybe van der Waals radius is more fundamental than people think,’ Tkatchenko says – it’s not after all just arbitrary where we choose to assign a cutoff in the decaying electron density.
We must learn to live with imprecise, statistical measures
Santiago Álvarez of the University of Barcelona in Spain
That possibility is supported by the fact that, empirically, Tkatchenko and his colleagues found the van der Waals radii for atoms such as the noble gases (which are not complicated by the formation of bonds) seem to be closely correlated with their polarisability – an objectively measurable quantity. Specifically, they found that the polarisability is proportional to the seventh power of the van der Waals radius.9
Why should this be? One way of thinking about the problem is that the apparent size of an atom depends on how its cloud of electrons interacts electromagnetically with the surrounding vacuum – which, in quantum field theory, manifests as a sea of virtual photons that the atom itself excites. Tkatchenko and colleagues have argued theoretically that the van der Waals boundary sits where the electric field due to the electrons becomes equal to that due to the virtual photons surrounding it.10 That’s not, then, an arbitrary choice, but one that is defined by the underlying physics: it’s where the attractive interactions due to the ubiquitous van der Waals forces (arising from correlations in electron density fluctuations) match the repulsive interactions caused by the Pauli exclusion principle, which says that electrons with the same quantum numbers can’t occupy the same region of space. ‘Underlying this is a deeper connection between the chemistry and the physics,’ Tkatchenko says.
Getting closer
The very notion of a covalent radius of an atom is inevitably idealised, because we can’t think of atoms in molecules as being perfectly spherical anyway. Bader suggested a way of carving up the electron density in molecules to give a sense of the size of atoms in molecules by looking for demarcating topological features like saddle points in the density map. But various other methods of assigning a value of covalent radius have been proposed, such as looking at the relation to electronegativity,11 ionization energy,12 or polarizability.13 Theoretical chemists Pekka Pyykkö and Michiko Atsumi of the University of Helsinki, Finland, have calculated single-bond covalent radii based on the distance of the atom from some probe atom or chemical group, such as hydrogen or methyl, to which it is bonded.14
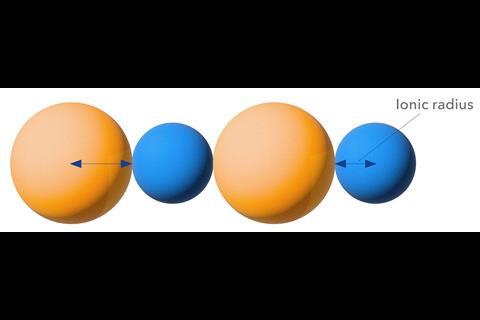
Ionic radii, meanwhile, have typically been calculated from crystal structures, assuming that the ions sit in contact so that the sum of the radii is equal to the distance between nuclei. They can also be derived from first principles using, say, density-functional theory with a particular choice of cutoff density.3 A widely used dataset of ionic radii15 are those published in 1969 by inorganic chemists Robert Shannon and Charles Prewitt of the du Pont Chemical Company in Wilmington, Delaware, US (which Shannon revised in 1976) based on crystallographic data, which recognised that the effect size of ions can depend on their coordination numbers and spin states. But it’s not clear that the ionic radii appropriate to the solid state are also the right ones to use in solution – and besides, these measures are complicated by the fact that some ionic bonds have a degree of covalency.
In theory these different types of interaction should connect to one another smoothly. At a large separation, two hydrogen atoms feel only the van der Waals force. But if you bring them gradually closer together, the interaction will gradually change into a covalent bond. ‘At the moment we don’t have a theory for this,’ says Tkatchenko. We might use a Morse potential (exponential in the distance between nuclei) to describe the covalent bond, and a Lennard-Jones potential (an inverse power law in distance) for the van der Waals forces – ‘but we don’t have a theory that connects the two’. He suspects this is an area where AI might help us to identify the general principles needed to bridge the gap.
Álvarez has suggested incorporating bonding interactions smoothly into the picture of the van der Waals atom by representing them in terms of interpenetration of ‘van der Waals crusts’.16 This penetration index, he says, may range from 0% when the van der Waals crusts just touch each other to 100% when they overlap all the way to the edge of the valence sphere, so that the atoms are fully covalently bonded. Such a scheme, he says, can be used to describe interactions ranging from the very weak van der Waals complexes of noble gases (typically less than 15% penetration) to interlayer interactions in two-dimensional van der Waals solids, hydrogen bonds (from 0% or even negative to about 90%), 1- and 3-electron s bonds (as in H2+ say; typically around 50–80%), and electron-deficient bonds (as in diborane B2H6). By this measure some very strong bonds, such as Cr–Cr quintuple bonds, can reach as much as 150% penetration.
There is, however, plenty more to be done to integrate notions of atomic size and chemical bonding. ‘For van der Waals radii things have become pretty much settled in the past five years,’ says Tkatchenko. But as far as other measures of atomic size relevant to other types of bonding, particularly covalent and ionic, are concerned, this is still ‘ongoing work’, he says.
It seems then that the question: how big are atoms? while not having a single and well-defined answer, is not about purely arbitrary choices either. There are measures of size that appear to have physical meaning in particular contexts, and we now have some idea of what those are and how to determine them. In the end, though, ‘we must learn to live with imprecise, statistical measures of atomic sizes,’ says Álvarez. But after all, isn’t learning to live with the imprecise what so much of chemistry is about?
References
1 R F W Bader, W H Henneker and P E Cade, J. Chem. Phys., 1967, 46, 3341 (DOI: 10.1063/1.1841222)
2 R J Boyd, J. Phys. B: At. Mol. Phys., 1977, 10, 2283 (DOI: 10.1088/0022-3700/10/12/007)
3 M Rahm, R Hoffmann and N W Ashcroft, Chem. Eur. J., 2016, 22, 14625 (DOI: 10.1002/chem.201602949)
4 A Bondi, J. Phys. Chem., 1964, 68, 441 (DOI: 10.1021/j100785a001)
5 M Mantina et al, J. Phys. Chem. A, 2009, 113, 5806 (DOI: 10.1021/jp8111556)
6 A Alibakhshi and B Hartke, Phys. Rev. Lett., 2022, 129, 206001 (DOI: 10.1103/PhysRevLett.129.206001)
7 A Alibakhshi and L Schäfer, ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-89wth
8 J Charry and A Tkatchenko, J. Chem. Theor. Comput., 2024, 20, 7469 (DOI: 10.1021/acs.jctc.4c00784)
9 A Tkatchenko, D V Fedorov and M Gori, J. Phys. Chem. Lett., 2011, 12, 9488 (DOI: 10.1021/acs.jpclett.1c02461)
10 D V Fedorov, M Sadhukhan, M Stöhr and A Tkatchenko, Phys. Rev. Lett., 2018, 121, 183401 (DOI: 10.1103/PhysRevLett.121.183401)
11 M Rahm, P Erhart and R Cammi, Chem. Sci., 2011, 12, 2397 (DOI: 10.1039/d0sc06675c)
12 P Politzer, P Jin and J S Murray, J. Chem. Phys., 2002, 117, 8197 (DOI: 10.1063/1.1511180)
13 R Khatiwada and P Li, J. Phys. Chem. A, 2025, 129, 23, 5118 (DOI: 10.1021/acs.jpca.5c02786)
14 P Pyykkö and M Atsumi, Chem. Eur. J., 2009, 15, 186 (DOI: 10.1002/chem.200800987)
15 R D Shannon and C T Prewitt, Acta Cryst. B, 1969, 25, 925 (DOI: 10.1107/S0567740869003220)
16 J Echeverría and S Alvarez, Chem. Sci., 2023, 14, 11647 (DOI: 10.1039/d3sc02238b)














2 readers' comments