An artificial ligament that integrates more strongly with the host’s bone than the best clinical material has been created using intertwined carbon nanotubes. When the nanotube ligament was transplanted into the legs of rabbits and sheep, they were able to run and jump normally once they had healed from the surgery.
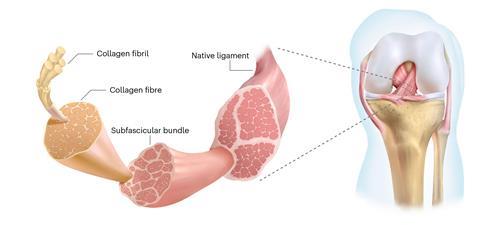
Ligaments connect bones together, and must withstand high longitudinal stress while remaining flexible. Natural ligaments comprise a rope-like hierarchy of nanometre-scale fibrils of collagen intertwined to form micrometre-scale fibres that are twisted into subfascicular bundles. When ligaments fail completely, finding replacements is challenging. A surgeon’s first choice is usually harvesting a tendon from the patient’s own body, but this can cause problems at the donor site. Alternatively, a cadaver can be used, but this poses potential – albeit manageable – risks of rejection or disease transmission, as well as restricted availability.
Producing artificial ligaments remains difficult as they need to be both strong and durable and form strong and stable interfaces with natural bones. The current state-of-the-art is helical PET fibres. Researchers at Fudan University in Shanghai synthesised multi-walled carbon nanotubes with diameters around 10nm by chemical vapour deposition and twisted these into hierarchical bundles analogous to those in native ligaments. In laboratory tests, the resulting fibres had superior stability to the PET fibres and mechanical properties close to biological ligaments.

The researchers then threaded the ends of the nanotube ligaments into holes drilled into the femurs and tibias of rabbits and sheep – the standard surgical procedure for reconstruction of the anterior cruciate ligament. Thirteen weeks later, the animals could run and jump normally. When they dissected the animals, they found that bone had grown around and even partially into the nanotube ligaments. In contrast, when they studied the bodies of animals implanted with PET ligaments, they found that the holes had widened around the artificial ligaments. The force required to pull the nanotube ligaments away from the bone was 1.4 times that required to detach the PET ligament and similar to the force required to remove native ligament. The researchers believe the greater ligament/bone integration may be partly because the microenvironment inside carbon nanotubes switches on cellular bone formation.
Experts in the field were cautiously complimentary. ‘The reason I like this research is that they show the functional recovery in the sheep,’ says Yunzhi Peter Yang at Stanford University in California. They now need to study the long term effects of the implants on the animals. One unanswered question, he notes, is whether the scaffold might stimulate bone growth along the ligament, impeding its movement.
Akhilesh Gaharwar at Texas A&M University agrees that ‘what stands out in this study is the observed [bone-tissue integration] in an anterior cruciate ligament replacement model’. However, ‘one of the key issues is the limited detail provided about the material properties of the carbon nanotube-based ligament, a factor that could significantly impact its clinical applicability and biocompatibility’. He also points out that manufacturing costs and potential regulatory issues are not discussed. ‘As such, while this study propels us towards an exciting direction in ligament replacements, it also underscores the need for further research to comprehensively evaluate the potential implications and efficacy of this new technology.’
















No comments yet